Chapter 26 Nucleosides, Nucleotides, and Nucleic Acids
1.13k likes | 1.38k Vues
Chapter 26 Nucleosides, Nucleotides, and Nucleic Acids. 26.1 Pyrimidines and Purines. Pyrimidines and Purines. In order to understand the structure and properties of DNA and RNA, we need to look at their structural components.

Chapter 26 Nucleosides, Nucleotides, and Nucleic Acids
E N D
Presentation Transcript
Pyrimidines and Purines • In order to understand the structure and properties of DNA and RNA, we need to look at their structural components. • We begin with certain heterocyclic aromatic compounds called pyrimidines and purines.
6 6 7 5 1 5 N N 1 N 8 4 2 2 9 N H 4 N N 3 3 Pyrimidines and Purines • Pyrimidine and purine are the names of the parent compounds of two types of nitrogen-containing heterocyclic aromatic compounds. Pyrimidine Purine
H NH2 H N N N H H2N H N H N N H Pyrimidines and Purines • Amino-substituted derivatives of pyrimidine and purine have the structures expected from their names. 4-Aminopyrimidine 6-Aminopurine
H H H H N N HO H O H N N H Pyrimidines and Purines • But hydroxy-substituted pyrimidines and purines exist in keto, rather than enol, forms. enol keto
O OH H N N N H N H N H N N H N H H keto Pyrimidines and Purines • But hydroxy-substituted pyrimidines and purines exist in keto, rather than enol, forms. enol
O O CH3 HN HN O O NH NH Important Pyrimidines • Pyrimidines that occur in DNA are cytosine and thymine. Cytosine and uracil are the pyrimidines in RNA. NH2 HN O NH Uracil Thymine Cytosine
NH2 O N N HN N NH NH H2N N N Important Purines • Adenine and guanine are the principal purines of both DNA and RNA. Adenine Guanine
O O CH3 CH3 H3C N N N HN N O O N N N CH3 CH3 Caffeine Theobromine Caffeine and Theobromine • Caffeine (coffee) and theobromine (coffee and tea) are naturally occurring purines.
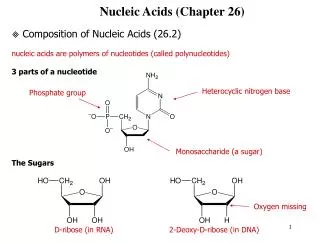
Nucleosides • The classical structural definition is that a nucleoside is a pyrimidine or purine N-glycoside of D-ribofuranose or 2-deoxy-D-ribofuranose. • Informal use has extended this definition to apply to purine or pyrimidine N-glycosides of almost any carbohydrate. • The purine or pyrimidine part of a nucleoside is referred to as a purine or pyrimidine base.
NH2 N N O HOCH2 O HO OH Table 26.2 • Pyrimidine nucleosides Cytidine Cytidine occurs in RNA; its 2-deoxy analog occurs in DNA.
O H3C NH O N HOCH2 O HO Table 26.2 • Pyrimidine nucleosides Thymidine Thymidine occurs in DNA.
O NH O N HOCH2 O HO OH Table 26.2 • Pyrimidine nucleosides Uridine Uridine occurs in RNA.
NH2 N N N N HOCH2 O HO OH Table 26.2 • Purine nucleosides Adenosine Adenosine occurs in RNA; its 2-deoxy analog occurs in DNA.
O N NH N NH2 N HOCH2 O HO OH Table 26.2 • Purine nucleosides Guanosine Guanosine occurs in RNA; its 2-deoxy analog occurs in DNA.
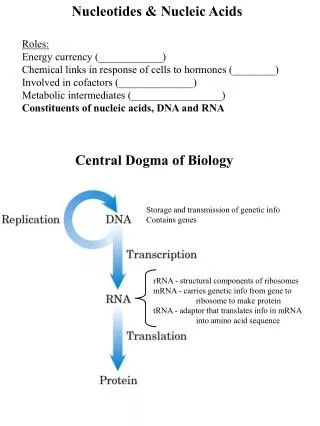
26.3Nucleotides • Nucleotides are phosphoric acid esters of nucleosides.
NH2 O N OCH2 HO N P 5' HO N N O 1' 4' 3' 2' HO OH Adenosine 5'-Monophosphate (AMP) • Adenosine 5'-monophosphate (AMP) is also called 5'-adenylic acid.
NH2 N N O O N N OCH2 HO O P P O HO HO HO OH Adenosine Diphosphate (ADP)
NH2 N N O O O N N HO OCH2 P O O P P O HO HO HO HO OH Adenosine Triphosphate (ATP) • ATP is an important molecule in several biochemical processes including: energy storage (Sections 26.4-26.5) phosphorylation
HOCH2 O O HO HO ATP + HO HO HO HO OH OH O (HO)2POCH2 ADP + ATP and Phosphorylation hexokinase This is the first step in the metabolism of glucose.
NH2 N N N N CH2 O O O OH P O HO cAMP and cGMP • Cyclic AMP and cyclic GMP are "second messengers" in many biological processes. Hormones (the "first messengers") stimulate the formation of cAMP and cGMP. Cyclic adenosine monophosphate (cAMP)
O N NH N NH2 N CH2 O O OH O P O HO cAMP and cGMP • Cyclic AMP and cyclic GMP are "second messengers" in many biological processes. Hormones (the "first messengers") stimulate the formation of cAMP and cGMP. Cyclic guanosine monophosphate (cGMP)
Bioenergetics • Bioenergetics is the thermodynamics of biological processes. • Emphasis is on free energy changes (DG). • When DG is negative, reaction is spontaneous in the direction written. • When DG is 0, reaction is at equilibrium. • When DG is positive, reaction is not spontaneous in direction written.
mA(aq) nB(aq) Standard Free Energy (DG°) • Sign and magnitude of DG depends on what the reactants and products are and their concentrations. • In order to focus on reactants and products, define a standard state. • The standard concentration is 1 M (for a reaction in homogeneous solution). • DG in the standard state is called the standard free-energy change and given the symbol DG°.
mA(aq) nB(aq) Standard Free Energy (DG°) • Exergonic: An exergonic reaction is one for which the sign of DG° is negative. • Endergonic: An exergonic reaction is one for which the sign of DG° is positive.
mA(aq) nB(aq) Standard Free Energy (DG°) • It is useful to define a special standard state for biological reactions. • This special standard state is one for which the pH = 7. • The free-energy change for a process under these conditions is symbolized as DG°'.
Hydrolysis of ATP ATP + H2O ADP + HPO42– • G°' for hydrolysis of ATP to ADP is –31 kJ/mol. • Relative to ADP + HPO42–, ATP is a "high-energy" compound. • When coupled to some other process, the conversion of ATP to ADP can provide the free energy to transform an endergonic process to an exergonic one.
–OCCH2CH2CHCO– + NH4+ +NH3 O O O O H2NCCH2CH2CHCO– + H2O +NH3 Glutamic Acid to Glutamine DG°' = +14 kJ Reaction is endergonic.
DG°' = –17 kJ O O O O H2NCCH2CH2CHCO– + HPO42– + ADP +NH3 Glutamic Acid to Glutamine –OCCH2CH2CHCO– + NH4+ + ATP +NH3 Reaction becomes exergonicwhen coupled to the hydrolysisof ATP.
O O O O O P OCCH2CH2CHCO– + ADP –O –O +NH3 Glutamic Acid to Glutamine –OCCH2CH2CHCO– + ATP +NH3 Mechanism involvesphosphorylation of glutamic acid.
H2NCCH2CH2CHCO– + HPO42– +NH3 O O O O O P OCCH2CH2CHCO– + NH3 –O –O +NH3 Glutamic Acid to Glutamine Followed by reaction of phosphorylated glutamic acid with ammonia.
Phosphodiesters • A phosphodiester linkage between two nucleotides is analogous to a peptide bond between two amino acids. • Two nucleotides joined by a phosphodiester linkage gives a dinucleotide. Three nucleotides joined by two phosphodiester linkages gives a trinucleotide, etc. (See next slide) • A polynucleotide of about 50 or fewer nucleotides is called an oligonucleotide.
free 5' end A T G free 3' end Fig. 26.1The trinucleotide ATG • phosphodiester linkages between 3' of one nucleotide and 5' of the next
26.7Nucleic Acids • Nucleic acids are polynucleotides.
Nucleic Acids • Nucleic acids first isolated in 1869 (Johann Miescher). • Oswald Avery discovered (1945) that a substance which caused a change in the genetically transmitted characteristics of a bacterium was DNA. • Scientists revised their opinion of the function of DNA and began to suspect it was the major functional component of genes.
Composition of DNA • Erwin Chargaff (Columbia Univ.) studied DNAs from various sources and analyzed the distribution of purines and pyrimidines in them. • The distribution of the bases adenine (A), guanine (G), thymine (T), and cytosine (C) varied among species. • But the total purines (A and G) and the total pyrimidines (T and C) were always equal. • Moreover: %A = %T, and %G = %C
Composition of Human DNA For example: • Adenine (A) 30.3% Thymine (T) 30.3% • Guanine (G) 19.5% Cytosine (C) 19.9% • Total purines: 49.8% Total pyrimidines: 50.1% Purine Pyrimidine
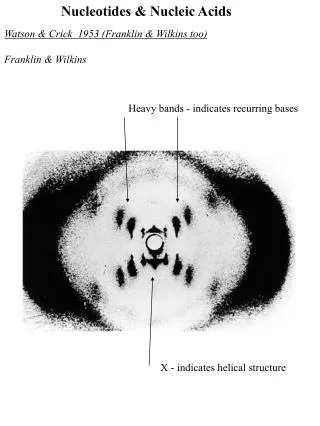
Structure of DNA • James D. Watson and Francis H. C. Crick proposed a structure for DNA in 1953. • Watson and Crick's structure was based on: •Chargaff's observations •X-ray crystallographic data of Maurice Wilkins and Rosalind Franklin •Model building
2-deoxyribose 2-deoxyribose A T Base Pairing • Watson and Crick proposed that A and T were present in equal amounts in DNA because of complementary hydrogen bonding.
Base Pairing • Watson and Crick proposed that A and T were present in equal amounts in DNA because of complementary hydrogen bonding.
2-deoxyribose 2-deoxyribose G C Base Pairing • Likewise, the amounts of G and C in DNA were equal because of complementary hydrogen bonding.
Base Pairing • Likewise, the amounts of G and C in DNA were equal because of complementary hydrogen bonding.
The DNA Duplex • Watson and Crick proposed a double-stranded structure for DNA in which a purine or pyrimidine base in one chain is hydrogen bonded to its complement in the other. • •Gives proper Chargaff ratios (A=T and G=C) • •Because each pair contains one purine and one pyrimidine, the A---T and G---C distances between strands are approximately equal. • •Complementarity between strands suggests a mechanism for copying genetic information.
Fig. 26.4 • Two antiparallel strands of DNA are paired by hydrogen bonds between purine and pyrimidine bases.