Vacuum Fundamentals
Vacuum Fundamentals 1 atmosphere = 760 mm Hg = 101.3 kPa 1 torr = 1 mm Hg vacuum range pressure range low 760 ~ 25 torr medium 25~ 10 -3 high 10 -3 ~ 10 -6 very high 10 -6 ~ 10 -9 ultrahigh 10 -9 ~ 10 -12

Vacuum Fundamentals
E N D
Presentation Transcript
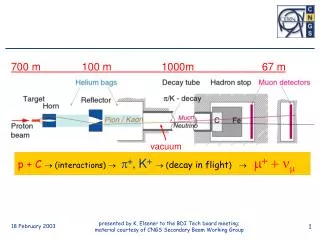
Vacuum Fundamentals 1 atmosphere = 760 mm Hg = 101.3 kPa 1 torr = 1 mm Hg vacuum rangepressure range low 760 ~ 25 torr medium 25~ 10-3 high 10-3 ~ 10-6 very high 10-6 ~ 10-9 ultrahigh 10-9 ~ 10-12 extreme high 10-12 ~ less Gas law: P=nkT, p: pressure in Pascal, n: molecular density (m-3), k: Boltzmans’ constant, T: temperature (k) Gas load: after initial pump down, gases continue to enter into chamber from leaks and outgassing True leaks: leaks from an infinite source such as air outside the chamber walls Trapped volumes: gases from trapped volumes (internal finite leaks), e.g. cracks in inside chamber walls, sandwiched gaps in screws and other internal fixtures
Adsorption: when exposed to ambient atmosphere, gases physisorbed or chemisorbed on the surface of Solids. These adsorbed layers consists mostly of water molecules with carbon oxides, oxygen, and nitrogen. (a monolayer forms in about half an hour even in 10-9 torr) Desorption: a strong function of temperature (temperature accelerates the desorption process) Permeation : -a gas migrates from the outer wall of a vacuum vessel to the inner wall (the gas adsorbs on the outer wall, and diffuses through the bulk of the solid, then desorbed from the interior wall) -For metal solids (not for glass, ceramic, and polymeric solids), hydrogen is one of the few gases that can measurably permeate metals Evaporation: release of molecular material from the same solid or liquid material to the gaseous phase Backstreaming: migration of molecules from the pump back into the pumped volume
Metals:- Better suited for vacuum systems then glasses, ceramics, and polymers (due to metal’s low absorption and permeation coefficient).- Alloys containing zinc, lead, cadmium, selenium, and sulfur should be avoided (high vapor pressures) - The most commonly used stainless steels are the 304 and 316 types 4 ways vacuum crossGlasses and Ceramics:Borosilicate glasses are used whenever baking temperature exceeds 350 ºC Fused silica is required for temperatures higher than 500 ºC. Glass viewport Materials
Polymers: the least desired material for vacuum applications due to their large porosity and their release of particles during compression
Del Seal 2-3/4 inch flange Kwik flange Copper gasket Vacuum components 4 inch gate valve
- Ionize gas, under strong electrical potentials (3~7 kV) accelerate them into a solid electrode. (swirling electrons in plasma ionizes incoming gas atoms and molecules while they are trapped in a strong magnetic field) - The swirling ions strike the chemically active cathode inducing sputter and are then pumped by chemisorption which effectively removes them from the vacuum chamber, resulting a net pumping action - capable to reach <10-11 mbar Ion pump
Diffusion pump -capable to reach 10-9 mbar -high speed, low cost
Cryogenic pump Trap gas by condensing them on a cold surface Depending on freezing and boiling points of gases (hydrogen will not condense but can be cryotrapped at 8K Can be operated with liquid nitrogen (boil at 77K) Over time, regeneration (exposure to vacuum and elevated temperature) is required to evaporate trapped gases
Turbomolecular pump fluid enters the pump near axis and rotor accelerates the fluid to high speed the fluid then passes through a diffuser which is a progressively enlarging pipe, which turns high kinetic energy into high pressure (hundreds of bar). can achieve 10-4 to 10-10 torr http://en.wikipedia.org/wiki/Turbopump
Positive displacement pump • Rotary vane pump • Diaphragm pump • Piston pump • Scroll pump • Screw pump • Wankel pump
Rotary vane pump a positive displacement pump
Diaphragm pump also called membrane pump
Uses of vacuum incandescent light bulb vacuum packing ultra-high vacuum used in study of atomically clean substrates (good vacuum Reserve atomic-scale clean surface) high to ultra-high vacuum removes obstruction of air, allowing particle beams to deposit or remove materials without contamination (e.g. CVD, physical vapor deposition, dry etching) electrical properties of vacuum make Transmission Electron Microscope (TEM) and vacuum tube (prevent filament from oxidation) possible Note: due to shorter wavelength than visible light, TEM can achieve 1,000,000x, whereas light microscopes are limited to 1000x SEM can reach 500,000x Fun with a vacuum: http://www.youtube.com/watch?v=QxD_-e-GkmY Vacuum tube: http://en.wikipedia.org/wiki/Vacuum_tube