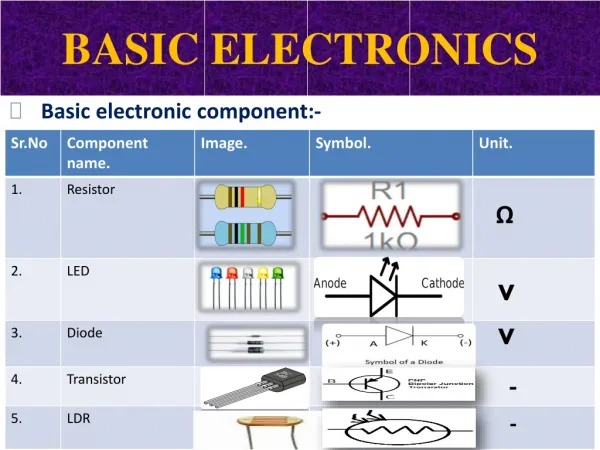
Basic Electronics
Basic Electronics. Basics in Mathmatics. Why is math important?. Read the article “ Math Mistakes in History: The Mars Climate Orbiter ” http://threesixty360.wordpress.com/2007/11/14/math-mistakes-in-history-the-mars-climate-orbiter / Explain what went wrong with the Mars Climate Orbiter.

Basic Electronics
E N D
Presentation Transcript
Basic Electronics Basics in Mathmatics
Why is math important? • Read the article “Math Mistakes in History: The Mars Climate Orbiter”http://threesixty360.wordpress.com/2007/11/14/math-mistakes-in-history-the-mars-climate-orbiter/ • Explain what went wrong with the Mars Climate Orbiter.
On 9/23/On 9/23/99, $125,000,000 Mars Climate Orbiter entered Mars’ atmosphere 100 km lower than planned and was destroyed by heat. 99, $125,000,000 Mars Climate Orbiter entered Mars’ atmosphere 100 km lower than planned and was destroyed by heat. 1 lb = 4.45 N “This is going to be the cautionary tale that will be embedded into introduction to the metric system in elementary school, high school, and college science courses till the end of time.”
What is scientific Notation? • Scientific notation is a way of expressing really big numbers or really small numbers. • It is most often used in “scientific” calculations where the analysis must be very precise.
Why use scientific notation? • For very large and very small numbers, these numbers can be converted into scientific notation to express them in a more concise form. • Numbers expressed in scientific notation can be used in a computation with far greater ease. • Mass of Proton: • .00000000000000000000000167 grams • Number of electrons passing by a point in a circuit: • 6250000000000000000 electrons per second
Scientific notation consists of two parts: • A number between 1 and 10 • A power of 10 N x 10x Are the following in scientific notation?
To change standard form to scientific notation… • Place the decimal point so that there is one non-zero digit to the left of the decimal point. • Count the number of decimal places the decimal point has “moved” from the original number. This will be the exponent on the 10. • If the original number was less than 1, then the exponent is negative. If the original number was greater than 1, then the exponent is positive.
Example 1 • Given: 289,800,000 • Use: 2.898 (moved 8 places) • Answer: 2.898 x 108
Example 2 • Given: 0.000567 • Use: 5.67 (moved 4 places) • Answer: 5.67 x 10-4
Practice • Use the link below to practice converting standard form to scientific notation. • Converting to Scientific Notation
To change scientific notation to standard form… • Simply move the decimal point to the right for positive exponent 10. • Move the decimal point to the left for negative exponent 10. (Use zeros to fill in places.)
Example 3 • Given: 5.093 x 106 • Answer: 5,093,000 (moved 6 places to the right)
Example 4 • Given: 1.976 x 10-4 • Answer: 0.0001976 (moved 4 places to the left)
Practice • Use the link below to practice converting scientific notation to standard form. • Converting to Standard Form
Even More Practice • Below is a list of links to games and activities all having to do with scientific notation. • http://www.aaamath.com/dec71i-dec2sci.html • http://janus.astro.umd.edu/cgi-bin/astro/scinote.pl • http://www.sciencejoywagon.com/physicszone/lesson/00genral/dectosci.htm
Now take the quiz to test your scientific notation skills! • Click on the link below to take the quiz and then use the answer key for the correct answers. • Quiz • Answers
I can rearrange equations to solve equations for specified variables. • What is the equation for solving for speed? • Can you rearrange the equation to find distance? • Can you rearrange the equation to find time?
Ohms law • defines the relationship between voltage, current and resistance. • These basic electrical units apply to direct current, or alternating current. • Ohm’s Law is the foundation of electronics and electricity. • This formula is used extensively by electricians. • Without a thorough understanding of “Ohm’s Law” an electrician can not design or troubleshoot even the simplest of electronic or electrical circuits. • Ohm established in the late 1820’s that if a voltage was applied to a resistance then “current would flow and then power would be consumed”.
Ohm's law magic triangle Resistance = R Voltage = E or V Current = I
If you know E and I, and wish to determine R, just eliminate R from the picture and see what's left:
If you know E and R, and wish to determine I, eliminate I and see what's left:
if you know I and R, and wish to determine E, eliminate E and see what's left:
To understand electronics we must review basic electricity. What is Electricity? • Everything is made of atoms • There are 118 elements, an atom is a single part of an element • Atom consists of electrons, protons, and neutrons
Electrons (- charge) are attracted to protons (+ charge), this holds the atom together • Some materials have strong attraction and refuse to loss electrons, these are called insulators (air, glass, rubber, most plastics) • Some materials have weak attractions and allow electrons to be lost, these are called conductors (copper, silver, gold, aluminum) • Electrons can be made to move from one atom to another, this is called a current of electricity.
Surplus of electrons is called a negative charge (-). A shortage of electrons is called a positive charge (+). • A battery provides a surplus of electrons by chemical reaction. • By connecting a conductor from the positive terminal to negative terminal electrons will flow.
Voltage • A battery positive terminal (+) and a negative terminal (-). The difference in charge between each terminal is the potential energy the battery can provide. This is labeled in units of volts. Water Analogy
Current • Uniform flow of electrons thru a circuit is called current. WILL USE CONVENTIONAL FLOW NOTATION ON ALL SCHEMATICS
Resistance • All materials have a resistance that is dependent on cross-sectional area, material type and temperature. • A resistor dissipates power in the form of heat
Create a Table to organize the variables and specific characteristics of each.
Conversions of units • As a milliampere (milliamp or just mA) is 1/1000th of an ampere, we can convert mA to Amps by just dividing by 1000. Another way is to take the current in mA and move the decimal to the left three places to accomplish the division by 1000. Here's the scoop: • 275 mA / 1000 = 0.275 Amps • Note that the decimal in 275 is to the right of the 5, and it's written as 275.0 (with a 0 added to show where the decimal is). Moving the decimal to the left three places gets up to .275 Amps, but we usually hang a 0 in front of the decimal. • To convert Amps to milliAmps, just multiply by 1000 or move the decimal to the right three places. Just the opposite of what we did here to convert the other way.
Electrical Power • Power is the rate of using or supplying energy: • Power = Energy / Time • Power is measured in watts (W)Energy is measured in joules (J)Time is measured in seconds (s)
Electrical Power • Electronics is mostly concerned with small quantities of power, so the power is often measured in milliwatts (mW), 1mW = 0.001W. • For example an LED uses about 40mW and a bleeper uses about 100mW, even a lamp such as a torch bulb only uses about 1W. • The typical power used in mains electrical circuits is much larger, so this power may be measured in kilowatts (kW), 1kW = 1000W. For example a typical mains lamp uses 60W and a kettle uses about 3kW.
Electrical Power Besides this basic equation for power: P = I*V remember we also have Ohm’s Law: V = I*R . Challenge: Based upon the two equations, how is power related to resistance?
Power is the rate of using or supplying energy P Power = Energy time Power is measured in watts (W)Energy is measured in joules (J)Time is measured in seconds (s)