Membrane Structure
Membrane Structure. Creation of the first cell and membrane


Membrane Structure
E N D
Presentation Transcript
Membrane Structure • Creation of the first cell and membrane • The two most important elements of cellular life—the boundary that separates the “inside” and the “outside” of the cell—the membrane system; and the inheritance molecule—one that can duplicate itself and passed on to the next generation (what?). • The evolution of the membrane—from the single cell membrane in the first cell and the primitive cells like bacteria (prokaryotes) to more complicated membrane systems—not only the outside boundary but intracellular membranes that surrounds each compartment or organelle. Plasma membrane (PM), chloroplast envelopes, ER, Golgi, Mitochondria, Nucleus, … • Functional diversity—composition hold the key-- What makes the difference? Proteins! PM has transporters, receptors…Nuclear proteins; chloroplast and photosynthetic complexes…
Lipid bilayer Ancestor cell
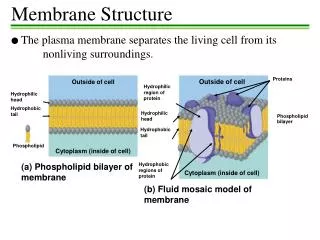
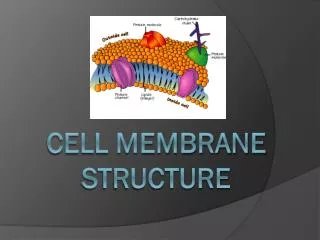
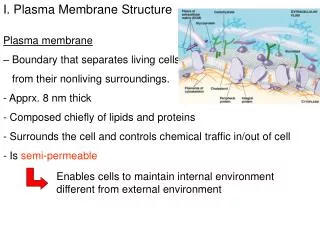
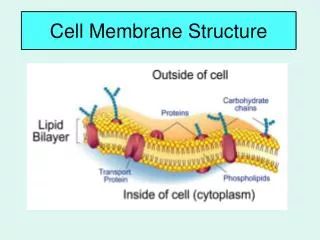
Lipid bilayer • 1) composition: phospholipids (50% or more) and other lipids: why choosing these materials? Chemical properties perfect for the purpose. Minors: Cholesterols and others. Amphapathic/ Amphaphilic molecules
2) Formation of bilayers: It is an automatic/spontenous process. How so? The chemical nature of phospholipids and other amphapathic molecules (such as detergents) allows interaction with water and formation of two structures in water solution: the micelles and the bilayer vesicles water This is like a tiny oil droplet (w/ polar head facing the water and the non-polar tails facing each other and away from water) water This is like a pita-bread w/ water both inside and outside (more stable than the micelle)
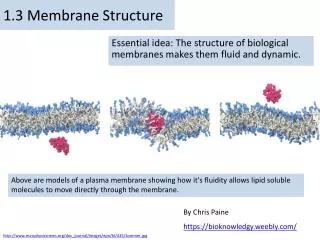
3) Dynamics of lipid bilayer • Mobility: children blow bubbles in the air—if you look carefully the surface of the bubbles “moves”. The molecules in the lipid bilayer move just like those detergent molecules in the “bubbles”. This property is called “mobility” of the bilayer. • There are several types of movements: lateral diffusion (lateral movement in the same layer), tail flexion (hydrophobic tails kick around inside the layer), rotation (the molecule turns around inside the same layer); vertical bobbing; and exchange between the two layers (flip-flop—requires enzymes to occur).
B) Fluidity, melting point, and phase transition of lipid bilayers The mobility of molecules in the lipid bilayer determines the “fluidity”. Like other fluid/liquid, lipid bilayers also change between liquid and solid state—phase transition. The temperature at which the transition occurs is the Melting point of a particular lipid bilayer.
What determines melting point of a lipid bilayer? This Tm, like Tm of water and other things is a “constant” for a particular matter. It is determined only by its chemical composition. As a lipid bilayer consists of many molecules, a number of factors affect its Tm. The composition of the lipids: the length and the saturation of the fatty acid chain/tails, the content of cholesterols are major factors---the composition of the cell membranes are dynamic and regulated based on the developmental and environmental responses: An example on how plant cells regulate its membrane Tm: During winter time or when temperature suddenly drops, some plants can change the composition of their membranes. One way is to decrease the saturation of the membrane fatty acids—to decrease Tm so that the membrane can be “fluid” at even lower temperature. This is done by increasing he level of enzyme activity of desaturase. Low temperature---activation of genes encoding desaturase---more mRNA—more protein—more activity---more unsaturated fatty acid---Tm drops.
How to measure membrane property such as fluidity and function of a particular membrane component? Model systems: liposome (bilayer sphere): used for measuring mobility of a lipid molecule, deliver drugs and DNA into the cells etc… planar membrane (bilayer within a plastic support): study permeability and transport activity of the membrane and a membrane protein (channel or transporter)
C) Asymmetry of lipid bilayers: the compositions of the two layers (leaves) of the bilayer are different—asymmetrical. This also refer to other components such as proteins in the membrane.
4) Membrane proteins—what determine the function of the membrane • Peripheral and integral proteins: also called extrinsic and intrinsic membrane proteins: referring to the way a protein is associated with the lipid bilayer--- defined based on experimental procedures to extract the proteins—some can be extracted by high salt and extreme pH conditions and are peripheral/extrinsic proteins (they were loosely bound to the surface of the membrane through salt or ionic interactions); those that can be exacted only by breaking the membrane apart using detergents are integral/intrinsic proteins (bound tightly and often span membrane bilayer)----see the above picture • Molecular and covalent interactions: Non-covalent bonds such as H-bond, ionic interaction, and van der Waals. The hydrophobic/van der Waals interaction is quite strong and is the major force for the integral proteins to interaction with the hydrophobic tails (interior of the membrane lipid bilayer), the H-bond and ionic interactions are weaker and usually used for peripheral proteins to associate with the surface of the membranes.
Covalent association: proteins may be modified by lipid or hydrocarbon chains so that he protein can be inserted into the membrane through this hydrophobic chain/tag.
3) Proteins form complexes in the membrane Ion channels, electron carriers in the mitochondria/chloroplast, the nuclear pore complexes etc… Multiple subunits and complicated assembly processes poorly understood. 4) Proteins move along with the membrane lipids—fluidity/mobility 5) Asymmetry: as mentioned earlier.