Understanding Nuclear Reactions: Stability and Radioactive Decay Explained
Nuclear energy involves unstable isotopes transitioning to stability by altering their nucleus, releasing substantial radiation energy in the process. Radioisotopes, which possess unstable nuclei, rely on the neutron-to-proton ratio for stability. Excess neutrons contribute to instability, resulting in energy loss through radiation during radioactive decay. There are three primary decay types: Alpha decay (ejecting helium nuclei), Beta decay (neutron transforms into a proton and an electron), and Gamma decay (emission of electromagnetic radiation). This process continues in decay series until a stable isotope is formed.

Understanding Nuclear Reactions: Stability and Radioactive Decay Explained
E N D
Presentation Transcript
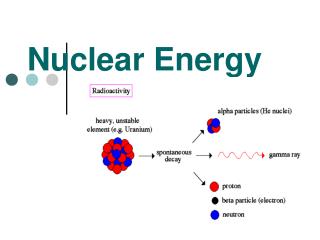
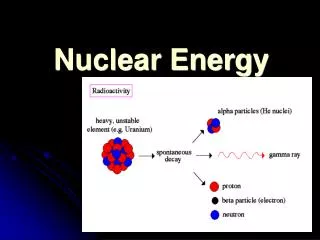
Nuclear Reactions- when unstable isotopes gain stability by making changes in the nucleus. • These changes are followed by large amounts of radiation energy
Radioisotopes-isotopes that have unstable nuclei. • *Stability depends on ratio of neutrons to protons. • *TOO many Neutrons leads to unstable nuclei. • *Lose energy by giving off radiation during radioactive decay.
3 Types: • Alpha ()(42He)-Helium nucleus(2 protons +2 neutrons) is ejected from source • Beta ()( 0-1e)-fast moving electrons formed from decomposition of a neutron. • Gamma ()-Electromagnetic radiation
Alpha: • 23692U232 90Th + 42He • When you lose alpha particle, Atomic # lowered by 2 and massby 4
Beta: • 10n 1 1H + 0 -1e • neutron breaks into proton and electron. Electron ejected and proton stays in nucleus.
Gamma: • 23290Th 22888Ra + 42He + • 23290Th 23291Pa + 0-1e + • Gamma often emitted with alpha or Beta radiation.
Decay Series • A series of radioactive nuclides produced by successive radioactive decay until a stable nuclide is reached. • Parent nuclide- heaviest nuclide of each decay series • Daughter nuclide- nuclides produced by the decay of the parent nuclides