Atoms and Compounds
Explore the evolution of atomic theory from Democritus to Dalton, understanding the nature of matter, elements, compounds, and chemical reactions. Discover the fundamental principles that govern the behavior of atoms in compounds.

Atoms and Compounds
E N D
Presentation Transcript
Atoms and Compounds Dalton’s Atomic Theory
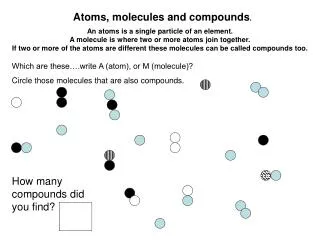
3.2Atoms and Compounds • Democritus (450 B.C.) – proposed that matter was not infinitely divisible; that matter could be broken down into tiny particles, which were not divisible. • These particles were referred to as atomos. • Atom – smallest particle of an element that retains the chemical identity of that element.
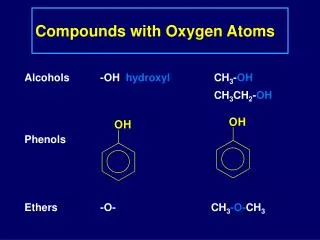
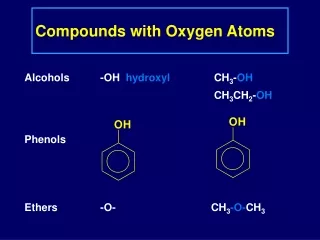
3.2 Atoms and Compounds Antoine Lavoisier – (late 1700’s) used a balance to show that matter was not created or destroyed during chemical reactions – Law of Conservation of Matter (Mass). Joseph Proust (1799)- showed that compounds always contain exactly the same proportion of elements by mass– Law of constant composition a.k.a. Law of definite proportion For example: The mass of water (H2O) always contains exactly 88.9 % oxygen and 11.1 % hydrogen.
Dalton’s Atomic Theory • John Dalton – (early 1800’s) proposed the atomic theory of matter • Compound 2 has exactly twice as much oxygen as Compound 1
Dalton’s Atomic Theory This can be explained in terms of atoms. Compound 1: CO (Carbon monoxide) Compound 2: CO2 (Carbon dioxide) Law of Multiple Proportions When 2 or more elements form a series of compounds, the ratios of the masses of elements can be reduced to small whole #’s
Dalton’s Atomic Theory • Each element is made up of tiny particles called atoms. • The atoms of a given element are identical; the atoms of different elements are different in some fundamental way • Chemical compounds are formed when atoms of different elements combine with each other. A given compound always has the same relative numbers and types of atoms • Chemical reactions involve reorganization of the atoms – changes in the way they are bound together. Atoms are not changed, created, or destroyed in a chemical reaction.