Solids and Phase Changes
E N D
Presentation Transcript
Properties of Solids • Molecules are tightly packed • All IMF forces exert stronger effects in solids • Forces hold particles in fixed positions • Only vibrational movement around fixed points • More order of particles due to stronger IMFs.
Types of Solids • Crystalline—consist of crystals • Crystal—particles are arranged in a orderly, geometric, repeating pattern. • Amorphous—particles are arranged randomly.
Properties of Solids • Definite shape and volume • Crystalline solids—particles packed into crystal lattice • Amorphous—when broken, maintain a definite shape but do not exhibit a distinct geometric shape. • Glass—after being molded, shatters in irregular shapes
Properties of Solids • Definite Melting Point for crystalline solids • Melting—solid changes phase to a liquid by addition of energy as heat. • Melting point—temperature at which solid becomes liquid • KE of particles overcomes attractive forces • Amorphous solids = no definite melting point • Flow over range of temps • Particles are arranged randomly like a liquid.
Properties of Solids • High incompressibility • High density • Low rate of diffusion—millions of times slower than liquids or gases.
Crystalline Solids • Crystal structures—used to show arrangements of particles in a crystal lattice. • Unit cell—shows smallest unit of the repeating pattern of entire crystal
Crystalline Solids • 4 types • Ionic • Contains positive and negative ions in a repeating pattern. • Hard, brittle, high MP, good insulators • Ex. MgCl2
Crystalline Solids • Covalent network • Each atom is covalently bound to the neighboring atom • Hard, brittle, nonconductors or semiconductors • Ex. SiO2, Graphite, Diamond,
Crystalline Solids • Metallic • Metals held together by sea of electrons • Covalent molecular • Individual molecules held together by IMFs • Low MP, soft, good insulators
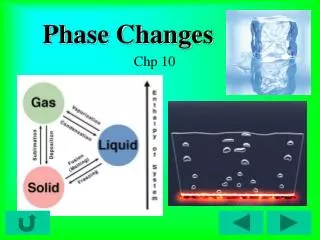
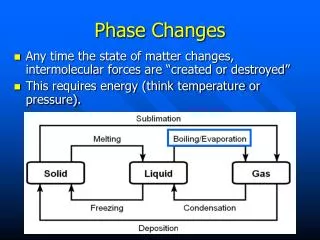
Changes of State • Phase change—any change from one state of matter to another. • Requires energy transfer to occur • Energy flows from higher energy particles to lower energy particles. • Intermolecular forces (IMF’s) are also affected.
Processes that take energy • Melting—changing from a solid to a liquid state • Energy change? • IMF’s? • Sublimation—change of state from a solid directly to a gas. • Vaporization—change of state from a liquid to a gas.
Phases releasing energy • Condensation—gas changes to liquid state • Energy change? • IMF’s? • Deposition—gas goes directly to a solid • Same process as condensation, except on much cooler surfaces • Freezing—liquid to solid
Equilibrium • Condition in which two opposing changes occur at equal rates in a closed system.
Phase Diagrams • Phase diagram—graph in which pressure versus temperature is used to show the conditions of phase changes for a specific substance. • Triple Point—point at which all three states of matter coexist. • Critical point—point of critical temp and critical pressure. • Critical temp—temperature above which the substance cannot exist as a liquid • Critical pressure—lowest pressure that substance can exist as a liquid.