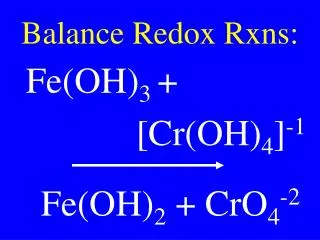Understanding Electrochemical Cells: Fundamentals and Applications in Battery Technology
170 likes | 307 Vues
Electrochemical cells are crucial for powering a variety of portable electronic devices, from laptops to mobile phones. A half-cell consists of a metal in contact with its ion solution, connected by a salt bridge and wire to allow electron flow. This potential difference can be measured, revealing the electrochemical series and the feasibility of reactions. Understanding these principles helps in predicting reaction outcomes using the anticlockwise rule and calculating E° reaction values. This knowledge is essential for advancements in battery technology.

Understanding Electrochemical Cells: Fundamentals and Applications in Battery Technology
E N D
Presentation Transcript
Redox: Electrochemical Cells Summary
Introduction- Electricity on the Move Batteries are vital for a whole variety of portable electric and electrical gadgets: laptop computers, mobile phones, personal stereos and camcorders
1 A half cell consists of….. A metal in contact with a solution of its own ions
2 The salt bridge connects…… The ions in the two half cells
3 The wire connects the two different half cells allowing….. The flow of electrons
4 Half cells develop electrical potential because of….. The tendency of all metals to give away electrons
5 If two half cells are connected by a votmeter and salt bridge…. Their potential difference(e.m.f) can be measured
6 If the other half cell is a standard hydrogen elctrode, the measured emf is called… Eo(E standard)
7 Eovalues are measured at standard conditions which are ….. 1mol/dm3 solutions, 100 Kpa pressure and 298K temperature
8 Eovalues for half cells are listed in order, with the most negative(the best electron releasers) at the top, to give….. The electrochemical series
9 The electrochemical series can be extended to systems other than metal/metal ion by….. Making electrical contact with a platinum electrode
10 Half cells high in the electrochemical series can transfer electrons to ….. Half cells lower in the list
11 The feasibility of reactions can be predicted by ….. The anticlockwise rule
12 The feasibility of reactions can also be predicted by…. Calculating Eoreaction
13 This is done by ….. Adding together two half reactions and their Eo values
14 In order for the electrons in the half reactions to cancel out …. one of the half reactions must be reversed (and thus the sign of itsEo value changed )
15 If the reaction is feasible…. Eoreaction is positive usually greater than 0.4V.