Defining the Atom
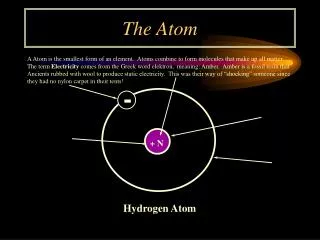
Defining the Atom. Democritus. 460B.C. – 570 B.C. Termed the name atom from the Greek word “atomos”. Philosopher If you break down an object you will eventually reach a point where you can not break it down any further. He called the smallest unit “atomos”. John Dalton.

Defining the Atom
E N D
Presentation Transcript
Democritus • 460B.C. – 570 B.C. • Termed the name atom from the Greek word “atomos”. • Philosopher • If you break down an object you will eventually reach a point where you can not break it down any further. He called the smallest unit “atomos”.
John Dalton Father of the “Atomic Theory” • All elements are made up of atoms. • Atoms of an element are identical. • Atoms of different elements combine together in whole number ratios (you will never see a ½ or 1/3, etc.) • i.e. H2O, CO2, C6H12O6, etc. • In chemical reactions, atoms are not changed, they are only rearranged. If you change atoms, it is nuclear and explosive.
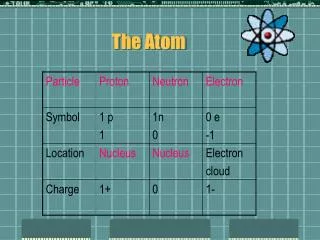
Structure of Nuclear Atom • Change in Dalton’s Atomic Theory is that atoms are divisible into subatomic particles: • Electrons (e-) • Protons (p+) • Neutrons (n0)
J.J. Thompson • Found electron by cathode ray tube.
Robert Millikan • Discovered the mass of the electron through an oil drop test. Mass of electron = 9.11 x 10-28 g or 0.0000000000000000000000000000911 g Too small ---- Insignificant ----Basically zero!
Food for Thought • J.J. Thompson discovered there was a negative particle called the electron. Robert Millikan discovered this negative particle has a very very small mass. • Thought: • If there is a negative, there must be a positive. • If electrons are so small (relatively no mass), what in the atom makes up its mass?
Eugene Goldstein • Discovered the positively charged proton and its mass of 1 amu. • Amu = atomic mass unit
James Chadwick • Discovered the neutron with no charge and its mass of 1 amu.
Plum Pudding Model • Thompson’s Atomic Model • Protons and Electrons are randomly dispersed throughout.
Ernest Rutherford • Gold Foil Experiment • Shot positive helium atoms through a thin gold foil. Lots of the helium cations went through and only a few deflected back. • Found there was a concentration of positive (protons). He called this concentrated spot the nucleus.
Conclusions • Nucleus is small. • Nucleus is dense. • Nucleus is positively charged. • Atom is mainly empty space. Thus, we no longer have the Plum Pudding Model, instead it looks like this:
Three Subatomic Particles • Neutron • Proton • Electron
Neutron • Has a mass of 1 • Has no charge (0) • Found in the nucleus
Proton • Has a mass of 1 • Is positively charged (+1) • Determines the identity of the atom • Found in the nucleus
Electron • Has NO Mass • Is negatively charged (-1) • Found on the outside of the nucleus • Atoms gain and lose electrons
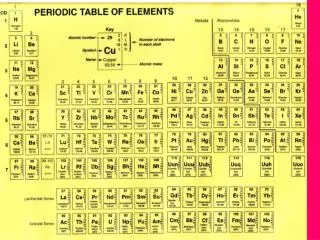
Element • Elements are made of atoms with the same number of protons. • There are many elements identified and scientists have placed them on the periodic table.
How do we read the Periodic Table? • Atomic Number • Number of Protons • Atomic Mass • Number of Protons and Neutrons
How do we figure out the number of neutrons? • Take the atomic mass and subtract the atomic number. Mass (neutrons + protons) – Atomic Number (protons)
Let’s Try Together Atomic Weight – Atomic Number Atomic Weight = 12.01 ~ 12 Atomic Number = 6 12 - 6 = 6 Neutrons
Now You Try! What is the atomic mass? 137.327~137 What is the atomic #? 56 How many neutrons does Barium have? 137 – 56 = 81 Neutrons
Let’s Reassess Our Knowledge • What does the atomic number tell us? • Number of protons. • What does the atomic mass tell us? • Number of protons and neutrons. • Which subatomic particle determines the identity of the element? • Proton • Which subatomic particles have mass? • Neutrons and Protons • Which subatomic particle do atoms gain and lose most often? • Electrons
Ions • An ion is a charged atom that is formed when an atom gains or loses an electron. • Anion • A negatively charged atom • An atom gains an electron • It is gaining negatives, so it becomes negative • Cation • A positively charged atom • An atom loses an electron • It is losing negatives, so it becomes positively charged
Identifying the Type of Ions • Ca+2 • O-2 • U6+ • Sn4+ • N-3 Cation Lost Electrons 2 Gained Electrons 2 Anion Cation Lost Electrons 6 Cation Lost Electrons 4 Anion Gained Electrons 3
Isotopes • Atoms with the same number of protons, but a differentnumber of neutrons. • Same element with a different atomic mass.
Determining the Number of Neutrons • Remember: To find the number of neutrons, you must take the atomic mass and subtract the atomic number. • Another way to write the element: • Element Symbol – Atomic Mass • C - 12 • Determine the Neutrons in the Isotopes: • Li-6 • Li-7 6-3 = 3 Neutrons 7-3 = 4 Neutrons
Other Ways to Write • Carbon-12 • Carbon-13 • Carbon-14 • C • C • C C 12 Atomic Mass 6 13 Atomic Number 6 14 6
Average Atomic Mass • Why does the mass have numbers after the decimal? • Elements contain atoms with different masses. • Same number of protons, but different number of neutrons (isotopes of the same element).
What is Average Atomic Mass? • Average Atomic mass • Average of all atoms with the same number of protons. • Thus, it is the average of isotopes for that element. • Abundance • Amount of that isotope in nature. • Displayed in percentage.
How Do We Determine the Average Atomic Mass? • Write the abundance and the corresponding isotope mass. • Rewrite the percent abundance as a decimal by moving the decimal two places to the left. • Multiply abundance (decimal) of that isotope by the mass. • Repeat step 1-3 for all isotopes. • Add all the numbers together.
Lets Try Together • Calculate the average atomic mass of iron if its abundance in nature is 15% iron-55 and 85% iron-56. 15% iron-55 0.15 0.15 x 55 8.25 8.25 + 47.6 55.85 amu 85% iron-56 0.85 0.85 x 56 47.6 1. Write the percent abundance and corresponding isotope mass. 2. Rewrite the percent abundance as a decimal. 3. Multiply abundance (decimal) by the isotope mass. 4. Add the numbers together.
In-Class Practice #2 • What is the average atomic mass of silicon if 92.21% of its atoms have a mass of 27.977 amu, 4.07% have a mass of 28.976 amu, and 3.09% have a mass of 29.974 amu? 92.21% Si-27.977 .9221 .9221 x 27.977 25.7975917 25.7975917 1.1793232 + 0.9261966 27.903 amu 4.07% Si-28.976 .0407 .0407 x 28.976 1.1793232 3.09% Si-29.974 .0309 .0309 x 29.974 0.9261966 1. Write the percent abundance and corresponding isotope mass. 2. Rewrite the percent abundance as a decimal. 3. Multiply abundance (decimal) by the isotope mass. 4. Add the numbers together.
In-Class Practice #3 • Calculate the average atomic mass for neon if its abundance in nature is 90.5% neon-20 (19.922 amu), 0.3% neon-21 (20.994 amu), and 9.2% neon-22 (21.991 amu). 90.5% 19.922 0.905 0.905 x 19.922 18.02941 18.02941 0.062982 + 2.023172 20.116 amu 0.3% 20.994 0.003 0.003 x 20.994 0.062982 9.2% 21.991 0.092 0.092 x 21.991 2.023172
In-Class Practice #4 • Calculate the average atomic mass of chromium. 4.35% 49.946 0.0435 x 49.946 2.172651 83.8% 51.941 0.838 x 51.941 43.526558 51.996 amu 9.5% 52.941 0.095 x 52.941 5.029395 2.35% 53.939 0.0235 x 53.939 1.2675665
Now You Try on Your Own! • Independent Practice • On Average Atomic Mass