Angiogenesis
Angiogenesis . The main steps that occur in angiogenesis from preexisting vessels are listed below: 1- Vasodilation in response to NO and increased permeability of the preexisting vessel induced by vascular endothelial growth factor (VEGF)


Angiogenesis
E N D
Presentation Transcript
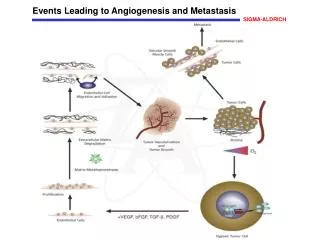
Angiogenesis • The main steps that occur in angiogenesis from preexisting vessels are listed below: 1- Vasodilation in response to NO and increased permeability of the preexisting vessel induced by vascular endothelial growth factor (VEGF) 2- Migration of endothelial cells toward the area of tissue injury 3- Proliferation of endothelial cells 4- Inhibition of endothelial cell proliferation and remodeling into capillary tubes 5- Recruitment of periendothelial cells (pericytes for small capillaries and smooth muscle cells for larger vessels) to form the mature vessel
Angiogenesis • New vessels formed during angiogenesis are leaky (incompletely formed interendothelial junctions and VEGF increases vessel permeability). This explains why granulation tissue is often edematous. • Growth Factors Involved in Angiogenesis: Several factors induce angiogenesis, but the most important are VEGF and basic fibroblast growth factor (FGF-2).
VEGF • VEGFs are dimericglycoproteins with many isoforms • Several agents can induce VEGFs, the most important being hypoxia. Other inducers are platelet-derived growth factor (PDGF), TGF-β, and TGF-α
FGFs • constitute a family of factors with more than 20 members, the best characterized are FGF-1 (acidic FGF) and FGF-2 (basic FGF). • are produced by many cell types • FGF-2 participates in angiogenesis mostly by stimulating the proliferation of endothelial cells. • It also promotes the migration of macrophages and fibroblasts to the damaged area, and stimulates epithelial cell migration to cover epidermal wounds.
Migration of Fibroblasts and ECM Deposition (Scar Formation) • It builds on the granulation tissue framework of new vessels and loose ECM that develop early at the repair site. • It occurs in two steps: • migration and proliferation of fibroblasts into the site of injury • deposition of ECM by these cells • The recruitment and stimulation of fibroblasts is driven by many growth factors, including PDGF, FGF-2, and TGF-β.
As healing progresses: • the number of proliferating fibroblasts and new vessels decreases • increased deposition of ECM. • Increased collagen synthesis (strength) • collagen synthesis by fibroblasts begins early in wound healing (days 3 to 5) and continues for several weeks, depending on the size of the wound • granulation tissue scaffold evolves into a scar (composed of spindle-shaped fibroblasts, dense collagen, fragments of elastic tissue, and other ECM components) • vascular regression (the highly vascularized granulation tissue transforms into a pale avascular scar)
Growth Factors Involved in ECM Deposition and Scar Formation • Many growth factors are involved in these processes, including TGF-β, PDGF, and FGF • TGF-β: • belongs to a family of homologous polypeptides • has many and often opposite effects, depending on the cell type and the metabolic state of the tissue. • In the context of inflammation and repair, TGF-β has two main functions:
TGF-β functions 1- is a potent fibrogenic agent. • It stimulates the production of collagen, fibronectin, and proteoglycans • It inhibits collagen degradation by both decreasing proteinase activity and increasing the activity of tissue inhibitors of proteinases known as TIMPs . • TGF-β is involved not only in scar formation after injury but also in the development of fibrosis in lung, liver, and kidneys that follows chronic inflammation. 2- TGF-β inhibits lymphocyte proliferation and can have a strong anti-inflammatory effect.
ECM and Tissue Remodeling • even after its synthesis and deposition, scar ECM continues to be modified and remodeled. • The outcome of the repair process is, in part, a balance between ECM synthesis and degradation • The degradation of collagens and other ECM components is accomplished by a family of matrix metalloproteinases (MMPs), which are dependent on zinc ions for their activity. • MMPs include: • interstitial collagenases, • gelatinases • stromelysins which degrade a variety of ECM constituents
Cutaneous wound healing • has three main phases: • inflammation, • formation of granulation tissue • ECM deposition and remodeling • Based on the nature of the wound, the healing of cutaneous wounds can occur by first or second intention.
Healing by First Intention • is the healing of a clean, uninfectedsurgical incision approximated by surgical sutures • is also referred to as primary union • The incision causes only focal disruption of epithelial basement membrane continuity and death of a relatively few epithelial and connective tissue cells. As a result, epithelial regeneration predominates over fibrosis. • The formed scar is small • There is minimal wound contraction.
Within 24 hours:neutrophilsmigration, fibrin clot formation • Within 24 to 48 hours: epithelial cells migration and proliferation along the dermis, basement membrane components deposition . The epithelial cells meet in the midline beneath the surface scab, yielding a thin but continuous epithelial layer. • By day 3:neutrophils replaced by macrophages, granulation tissue formation
By day 5:peak of neovascularization as granulation tissue fills the incisionalspace; Collagen fibrils become more abundant and begin to bridge the incision • During the second week:continued collagen accumulation and fibroblast proliferation; diminished leukocyte infiltrate, edema, and vascularity • By the end of the first month:the scar matures (connective tissue devoid of inflammatory cells and covered by an essentially normal epidermis). However, the dermal appendages destroyed in the line of the incision are permanently lost. The tensile strength of the wound increases
Healing by Second Intention • When cell or tissue loss is: • more extensive, such as in large wounds • abscess formation • ulceration • after infarction in parenchymalorgans • the repair process is more complex • also known as healing by secondary union
Secondary healing differs from primary healing in several respects • A larger clot or scab • Inflammation is more intense • Much larger amounts of granulation tissue are formed • involves wound contraction
Wound contraction • Within 6 weeks, for example, large skin defects may be reduced to 5% to 10% of their original size, by the process of wound contraction. • This process is the function of myofibroblasts, which are modified fibroblasts exhibiting many of the ultrastructural and functional features of contractile smooth muscle cells
Wound strength • When sutures are removed, usually at 1 week, wound strength 10% of that of unwounded skin, but this increases with time • by 3 months 70% to 80% of normal (but usually does not substantially improve beyond that point).
PATHOLOGIC ASPECTS OF REPAIR • Wound healing may be altered by a variety of influences: 1- Infection is the single most important cause of delay in healing 2- Poor nutrition: protein deficiency, vitamin C deficiency 3- Glucocorticoids (steroids): anti-inflammatory effects 4- Mechanical variables such as increased local pressure or torsion
5- Poor perfusion: arteriosclerosis and diabetes 6- foreign bodies such as fragments of steel, glass, or even bone
Keloid. A, Excess collagen deposition in the skin forming a raised scar known as a keloid. B, Thick connective tissue deposition in the dermis
Keloid • There is a heritable predisposition to keloidformation • is more common in blacks. • Tend to generate excessive granulation tissue that protrudes above the level of the surrounding skin and hinders re-epithelialization. • This is called exuberant granulation • requires cautery or surgical resection of the granulation tissue/ scar