ANTIBODY
ANTIBODY. Babitha Elias. DEFINITION Antibodies are substances which are formed in the serum or tissue fluids in response to an antigen. Antibodies react with antigen specifically and in an observable manner.


ANTIBODY
E N D
Presentation Transcript
ANTIBODY Babitha Elias
DEFINITION Antibodies are substances which are formed in the serum or tissue fluids in response to an antigen. Antibodies react with antigen specifically and in an observable manner. Immunoglobulin – proteins of animal origin, endowed with known Ab activity & for certain other proteins related to them by chemical structure.
Properties of antibodies Chemical nature of antibodies is globulin – immunoglobulin Constitute about 20 – 25% total serum proteins. Based on sedimentation studies – 7S (M.W – 1,50,000- 1,80,000 KD). Heavier antibodies – 19S globulins (M.W – 900,000) Kabat showed, on electrophoretic mobility, the antibodies belong to gammaglobulins
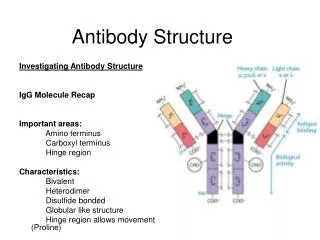
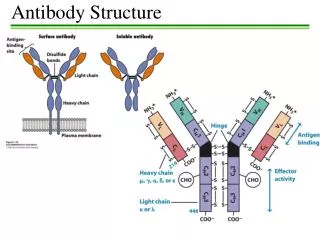
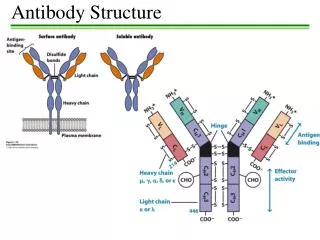
Structure of immunoglobulins Porter, Edelman and Nisonoff Antibody molecule consists of two identical heavy chains (H) and two identical light chains (L). Heavy chains are longer & light chains are shorter. Both chains are polypeptide in nature. 2 heavy chains are held together by disulphide bonds. Each light chain is attached to a heavy chain by disulphide bond.
The H chains are structurally and antigenically distinct in different classes of immunoglobulins. 5 different classes of immunoglobulins depending on the presence of heavy chain. Ig G – γ Ig M - μ Ig A - α Ig E - ε Ig D - δ
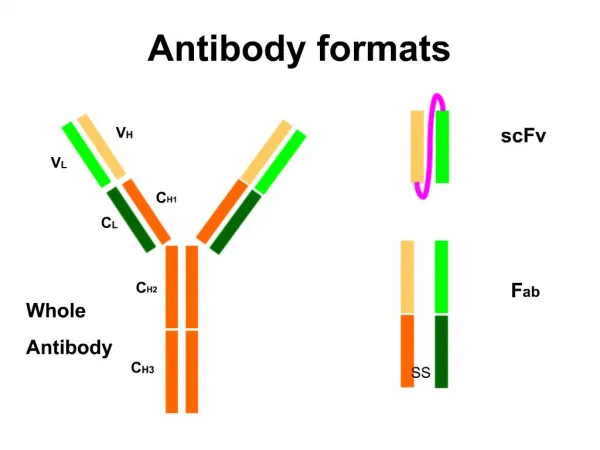
The L chains are similar in all classes of immunoglobulins. Present in two forms – kappa (K) and lambda (L). Effect of enzymes on Immunoglobulins Papain Digestion Papain can digest immunoglobulin molecule into 3 fragments – one Fc (Fragment crystallisable) and 2 identical Fab (fragment antigen binding) The 2 Fab fragments possess the antigen binding sites but the Fc fragment lacks the ability to bind antigen.
Each Fab fragment consists of a light (L) chain & a part of H chain Portion of H chain in Fab fragment – Fd region Fc fragment consists of both the H chains. It determines the biological properties of the Ig – complement fixation, placental transfer etc.
Pepsin digestion Pepsin cleaves immunoglobulin into 1 Fc portion & 2 Fab fragments held together in position. The Fab is bivalent & still it can precipitate with antigen – F(ab’)2 Pepsin degrades the Fc portion into smaller fragments.
H chains & L chains Consists of two portions – a variable region (V) & a Constant region (C). In the L chain, the 2 regions are of equal length, while in the H chain, the V region constitute only a fifth of the chain. Variable regions are in the amino terminus (NH2) and Constant region at carboxyterminus (COOH)
IMMUNOGLOBULIN CLASSES IMMUNOGLOBULIN G (Ig G) Major serum immunoglobulin – 80% of total amount. The normal serum concentration – 8-16 mg/ml. Molecular weight – 150,000 (7S) Half life – 23 days It can cross the placenta – Transplacental
It is distributed equally between the intravascular & extravascular compartments. Ig G appears late but persists long. It appears after the initial immune response. It participates in precipitation, complement fixation, & neutralization of toxin & viruses. Ig G binds to microorganisms & enhances phagocytosis.
Catabolism of Ig G – • When Ig G level is raised, the synthesis of Ig G against that particular Ag is catabolised rapidly & result in deficiency of particular Ab. Eg: myeloma & kala-azar. • In hypogammaglobulinaemia, Ig G antibody given for therapeutic purpose will be catabolised slowly. • Passively administered Ig G suppresses the homologous antibody synthesis by feed back mechanism. • 4 sub classes of Ig G – Ig G1, Ig G2, Ig G3, Ig G4 • It is protective against microorganisms which are active in the blood and tissues.
IMMUNOGLOBULIN A (Ig A) Second major serum immunoglobulin – 10- 13%. Normal serum concentration – 0.6-4.2 mg/ml. Half life – 6 – 8 days. Ig A occurs in two forms – Serum Ig A & Secretory Ig A. Serum Ig A is a monomeric 7S molecule.(MW: 160,000), While Ig A in the mucosal surfaces & secretions (Secretory Ig A, MW: 400,000) is a dimer. It is formed by two monomer units joined together by a glycoprotein – J chain
J chain – joins the 2 monomers at their carboxy terminals. Secretory Ig A & J chain are produced by the plasma cells. Secretory Ig A contains a secretory piece, S piece is synthesised by mucosal & glandular epithelial cells. The S piece protects Ig A from denaturation by bacterial proteases in sites such as intestinal mucosa. Ig A is present in secretions such as milk, saliva, tears, sweat, nasal fluids etc. It protects the mucus membranes against microorganisms..
Ig A cannot fix complement, but can activate alternative complement pathway. Ig A is synthesised locally by plasma cells. 2 subclasses of Ig A – Ig A1 & Ig A2
IMMUNOGLOBULIN M (Ig M) Ig M is a pentamer consisting of 5 monomers joined by a J chain. It constitutes about 5-8% of total serum concentration. Normal level – 0.5-2 mg/ml Half life – 5 days. Molecular weight – 19S (900,000-1,000,000) ‘Millionaire molecule’ Distributed intravascularly.
It is the earliest synthesised Ig by foetus in about 20 weeks of age. • It appears early in the infection before Ig G. Ig M Abs are short lived – presence indicates recent infection. • It cannot cross placenta – presence in new born indicates congenital infection. • It participates in agglutination, complement fixation, opsonisation & immune hemolysis • Ig M provides protection against blood invasion by microorganisms.
Ig M monomers appear on the surface of unstimulated B lymphocytes & act as receptors for antigens. • Two subclasses – Ig M1 & Ig M2.
IMMUNOGLOBULIN E (IgE) • Produced in the lining of respiratory & intestinal tracts. • Also known as “Reagin” • MW- 190,000 (8S) • Half life 2-3 days • Resembles Ig G structurally • Heat labile (Inactivated at 56 o C for 1 hr) • It has affinity for surface of tissue cells – Mast cells .(Homocytotropism)
Ig E mediates Type-I hypersensitivity • Responsible for asthma, hay fever, eczema, Prausnitz-Kustner (P-K) reaction. • Cannot cross the placental barrier. • Plays role in defense against parasitic infection.
IMMUNOGLOBULIN D (Ig D) • It resembles Ig G structurally. • Serum concentration – 0.03 mg/ml • Intravascular in distribution. • MW – 180,000 (7S) • Half life – 3days • It also act as receptors for antigen, like monomeric IgM . • 2 subclasses –IgD1, IgD 2
Role of different Ig classes IgG – Protects the body fluid IgA - Protects the body surface IgM – Protects the blood stream IgE - Mediates reaginic hypersensitivity IgD- Recognition molecule on the surface of B lymphocytes
ABNORMAL IMMUNOGLOBULINS • Apart from antibodies, other structurally similar proteins may be found in the serum in the following conditions. • Multiple myeloma • Heavy chain disease • Cryoglobulinaemia
Multiple myeloma • It is a plasma cell dyscrasia in which unchecked proliferation of one clone of plasma cells resulting in the excessive production of particular Ig. • Multiple myeloma involve plasma cells synthesizing any of the 5 classes of Ig. • Multiple myeloma involving IgM producing plasma cells – “Waldenstrom’s Macroglobulinaemia”
Excessive production of light chains – Bence Jones proteins. • It can be identified in urine – proteins get coagulated at 50oC but redissolve at 70oC Heavy chain disease • Abnormal heavy chains are produced in excess. This is due to lymphoid neoplasia.
Cryoglobulinaemia • It is a condition in which there is formation of precipitate on cooling the serum, which redissolves on warming. • Found in macroglobulinaemia, autoimmune conditions like SLE. • Most cryoglobulins consists of either Ig G or Ig M.