Ionic solids with formula Mx
Ionic solids with formula Mx. 201101021 박한솔. Contents. Basic crystal structure Metallic Crystals Structures of ionic solids with formula MX. Basic crystal structure. The primitive cubic structure - The most basic crystal structure, called simple cubic - All layers is identical

Ionic solids with formula Mx
E N D
Presentation Transcript
Ionic solids with formula Mx 201101021 박한솔
Contents • Basic crystal structure • Metallic Crystals • Structures of ionic solids with formula MX
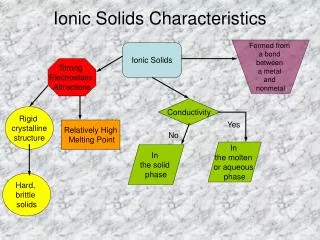
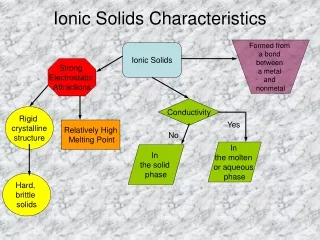
Basic crystal structure • The primitive cubic structure • - The most basic crystal structure, called simple cubic • - All layers is identical • - Total number of atoms in the unit cell (z) = 8 x 1/8 = 1 • - Coordination number (CN) is 6 • - Occupancy = 52.4% , 0.73r sphere
Basic crystal structure • 2. Body-Centered Cubic • Another sphere is added in the center of the simple cubic structure • CN = 8 • Occupancy is 68% • If the added sphere has the same radius as others it has 2.31r
Basic crystal structure • 3. Close-Packed Structures • Hexagonal Close Packing (hcp) • The result of an ABA structure • CN = 12 ( 6 in the same layer, 3 in layer above and 3 in layer below) • Occupancy is 74%
Basic crystal structure • 3. Close-Packed Structures • (2) Cubic Close Packing(ccp) or Face-Centered Cubic(fcc) • The result of ABC structure • CN = 12 • Occupancy is 74% • Unit cell is Face-centered cubic
Basic crystal structure • 4. Holes in Lattices • The packing of atoms into carious lattices produces interstices or “holes”, which may be filled to produce different compounds • Two different types of holes in close-packed structures
Basic crystal structure • (1) Tetrahedral holes • CN = 4 • Radius of the largest sphere fit in the tetrahedral holes = 0.225r • (2) Octahedral holes • CN = 6 • Radius of the largest sphere fil in the octahedral holes = 0.414r
Metallic crystals • Most metals crystalize in body-centered cubic(bcc), cubic close-packed(ccp), and hexagonal close-packed structures(hcp) • Changes in pressure or temperature can change metallic crystals • The sizes and packing of atoms are somewhat variable
Metallic crystals • Properties of Metals • High conductivity ( low resistance) to the passage of electricity and of heat • Some are soft and easily deformed by pressure or impact, or malleable (Cu, fcc) • Some are hard and brittle (Zn, hcp) (rare) • Boding is non-directional • Dislocation
Structures of ionic solids with formula MX • ※ the relative sizes of the atoms or ions • The radius ration(r+/r-) • Small cations can fit in the tetrahedral or octahedral holes of a close-packed anion lattices • Lager cations can if in the octahedral holes, but not in tetrahedral holes
Structures of ionic solids with formula MX • ※ the relative numbers of cations and anions • Formula M2X will not allow a close-packed anion lattice and occupancy of all the octahedral holes by the cations
Structures of ionic solids with formula MX • Caesium chloride structure (CsCl) • Simple primitive cubic of Cl • Cs in the centers • CN = 8 (both) • - CsCl, CaS, TlCl, CsCN
Structures of ionic solids with formula MX • (2) Sodium chloride (NaCl) • Known as rock salt • Unit cell is cubic • Consist of two interpenetrating • face-centered arrays • CN = 6 • Usually with the anions larger than • the cations • Many alkali halides • LiCl, KBr, MgO, AgCl, TiO, NiO, ScN
Structures of ionic solids with formula MX • (2) Sodium chloride (NaCl)
Structures of ionic solids with formula MX • (2) Sodium chloride (NaCl) • Alternative way of viewing • this structure • The close packing of one of • the ions
Structures of ionic solids with formula MX • (2) Sodium chloride (NaCl)
Structures of ionic solids with formula MX • (2) Sodium chloride (NaCl) • The other way to see “NaCl”
Structures of ionic solids with formula MX • MO6 octahedral link through • A vertex : M2O11 • An edge : M2O10 • A face : M2O9
Structures of ionic solids with formula MX • NaCl structure -> NaCl6 octahedral sharing edges
Structures of ionic solids with formula MX • (3) Nickel arsenide (NiAs) structure • Arsenic atoms in identical close-packed layers • Nickel atoms in all the octahedral holes • of a hexagonal close-packed arsenic lattice • CN = 6 (both) • MX compounds • M = transition metal • X = Group 14, 15 or 16 • (Sn, As, Sb, Bi, S, Se, or Te)
Structures of ionic solids with formula MX • (4) Zinc blende or Sphalerite (ZnS) • CN = 4 • The most common zinc ore • The same geometry as diamond • if all the atoms were identical • Each in face-centered lattices • Each ions in a tetrahedral hole of the other latice • The copper halides (CuX), Zn, Cd, Hg with sulfides
Structures of ionic solids with formula MX • (5) Wurtzite (ZnS) • - Polymorph(동질이상) with zinc blende • CN = 4 • Much rare than zinc blends • Formed at higher temperature • Each in a tetrahedral hole of the other • lattice • Each ion forms a hexagonal close-pack • Lattice • - BeO, ZnO, and NH4F