Ionization Energy and Electron Affinity
E N D
Presentation Transcript
Ionization Energy and Electron Affinity Section 7.4-7.5
Objectives • Examine periodic trends in ionization energy • Examine periodic trends in electron affinity
Key Terms • Ionization energy • Electron Affinity
Ionization Energy • Ease at which electrons can be removed from an atom or ion • First ionization energy, I1, is the energy required to remove the first electron from neutral atom • Second ionization energy, I2, is the energy required to remove the second electron from a +1 ion • Greater the I, the more difficult to remove the electron
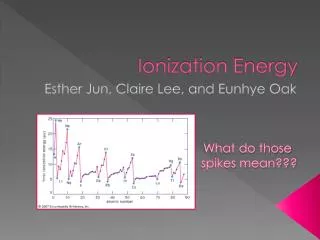
Trends of Ionization Energy • I1< I2 < I3 < I4 • With each removal, the ion becomes more positive • Strengthens nucleus’s hold on its electrons • Sharp increase in I when inner-shell electrons are removed • Table 7.2 on page 271
Periodic Trends in I1 • Within rows, I1 increases with atomic number • Slight irregularities • Within columns, I1 decreases with increasing atomic number • Representative elements show larger range of I1 values than transition metals • Transitions slowly increase across rows
Factors Affecting Trends • More energy is needed to remove an electron as: • Effective Nuclear Charge increases • Atomic Radius decreases
Irregularities • I does not always increases across rows • Decrease from Be to B Be: [He] 2s2 B: [He]2s2 2p1 • The most easily removed electron in B is that in the 2p orbital, higher in energy than the 2s electrons in Be • 2p electron of B needs less energy for total removal than does the 2s electron of Be
Irregularities • O is lower than N • N: [He]2s2 2p3 • O: [He]2s2 2p4 • Important difference in the way electrons are distributed • Hund’s rule: 3 e- in the 2p orbitals of a nitrogen atom all have the same spin, but e- are paired in one of the 2p orbitals of oxygen • Electrons try to stay as far apart as possible to minimize repulsion • Force of repulsion between these electrons is minimized to some extent by pairing electrons • Slightly easier to remove an electron
Electron Affinities • Energy change that occurs when electron are added to a gaseous atom • Measure of the attraction of an atom to an electron • Negative values (energy is released during process) • kJ/mol • More negative affinity = greater attraction
Trends in Affinities • Not as evident as that of ionization energy • Halogens have the greatest affinities (1 e- shy of a filled p subshell) • Noble gases have affinities of 0 • Group 2A and 5A have very little affinity
Homework • 7.35, 7.42, 7.44, and 7.46 on page 294