6.1 The Atom
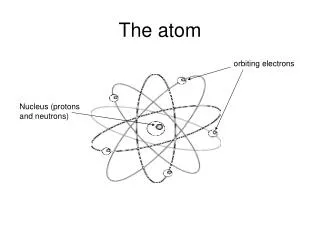
6.1 The Atom. Atomic Structure Nuclear Structure. 6.1.1. Describe a model of the atom that features a small nucleus surrounded by electrons. This is a simplified view of the atom known as the Rutherford model. History. Atom – the term atomos comes from Greek and means “indivisable”

6.1 The Atom
E N D
Presentation Transcript
6.1 The Atom Atomic Structure Nuclear Structure
6.1.1 • Describe a model of the atom that features a small nucleus surrounded by electrons. • This is a simplified view of the atom known as the Rutherford model
History • Atom – the term atomos comes from Greek and means “indivisable” • Originally thought to be the smallest parts of matter • The spheres to the right are the top layer of a gold sheet • Each sphere is one atom
History • 1803 – John Dalton proposed that atoms reacted with each other • 1897 – JJ Thompson discovered the electron with the invention of the cathod ray tube • Thompson's model was the plumb pudding version, where negative "corpuscles" were distributed though out positive “pudding”
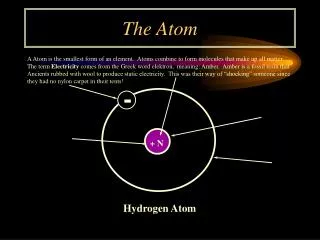
History • The Rutherford model had a positively charged nucleus, with the negatively charge electron orbiting the nucleus • In 1913 Niels Bohr added Quantum energy levels saying the electrons could only exist in certain orbits with definite energy • These models didn’t adequately explain the atom so in 1926 Schrödinger, citing DeBroglie’s wave model for mass, created his Model using the uncertainty principle
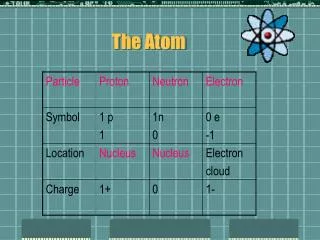
The Schrödinger Model • The nucleus contain the nucleons (Protons and Neutrons) with are made up of quarks • The electrons exist in electron clouds around the nucleus • All the components of the atom are made from elementary particles • Try this Atom Builder
6.1.2 • Outline the evidence that supports a nuclear model of the atom. • A qualitative explanation of the Geiger–Marsden experiment and its results is all that is required. • Also know as the Gold foil experiment, It was performed by Hans Geiger and Ernest Marsden in 1909 under the supervision of Rutherford
Evidence of the Nuclear model • The expected path for the “Plum pudding model” would be for an Alpha particle (He nucleus) to travel straight through or get absorbed • In reality some a-particles where reflected back • Rutherford said of the results "It was almost as incredible as if you fired a fifteen-inch shell at a piece of tissue paper and it came back and hit you". • Click here for a picture
6.1.3 • Outline evidence for the existence of atomic energy levels. • Students should be familiar with emission and absorption spectra, but the details of atomic models are not required. • Take a trip back to the Bohr model
If I only had an orbit • Imagine you are at the science museum and you see that yellow funnel thing, the spiral wishing well. • You know, the one you drop the penny in and watch it swirl around until in goes into the hole at the center • That is the fundamental problem with the Rutherford model of the atom, when the electron give off energy the loose momentum and thus should spiral into the nucleus
Bohr’s Solution • Bohr solved this by “allowing” the electrons to have discrete energies instead of continuous energies • The electron loses energy when it “drops” to a lower energy state • The emitted energy is the difference between the two states • This is evident in the emission and absorption spectra
Energy Levels • When an electron goes down in energy, the excess energy is released as a photon • In order to go up in energy, a photon must be absorbed • The energy associated with the transition is E = hf • h = 6.63 x 10-34 Js Photon out Photon in
Emission and absorption • Since certain wavelengths with be absorbed or emitted, a given atom will only produce or take in certain frequencies of light • Here are both spectrums for Hydrogen
6.1.4 Isotopes and neutrons • We already know there are protons and neutrons (nucleons) in the nucleus of the atom, but how do we know this? • The discovery of isotopes, and some very ingenious experiments brought the existence of the neutron to light
6.1.5 Explanation of terms • Nuclide - This name is given to a nucleus with a specific number of protons and neutrons • Here, A is the total number of nucleons, Z is the number of proton, and X is the atomic symbol • A nuclide is represented the following way:
Notation • This notation can be applied to proton, neutron, and electron as well • Isotopes are easy to identify using this notation • Common isotopes would be Carbon 14, Hydrogen 3, and Oxygen 18
Isotopes Hydrogen Deuterium Tritium • An isotope will have the same number of protons and a different number of neutrons • Isotopes will have the same chemical properties, but their physical properties will be slightly different
6.1.6 Atomic mass & number • Z denotes the number of protons in a nucleus, also called the atomic number • A denotes the total number of nucleons for an atom, so (Z + N = A) • N is the number of neutrons • If the atomic mass number changes the element changes
6.1.7 More forces • Obviously something has to hold together the nucleus • We learn in 5.1.7 positive charges repel each other, yet the protons in the nucleus are “glued” together • This “glue” comes from the strong nuclear force, which works at very short distances (10-15 m or less) • The atomic radius is written as R = 1.2 x A1/3 x 10-15, where A is the mass number.
Weak nuclear force • Since Neutrons and protons are made up of quarks there must be a force holding the quarks together as well • This force is the Weak nuclear force • A neutron will decay into a proton, an electron, and an anti-neutrino • This is called Beta decay • Click here to see more