Analyzing Mexico’s Pharmaceutical Landscape: Challenges and Opportunities
Explore Mexico’s pharmaceutical DNA, health system, and demographic trends. Learn about pricing policies, intellectual property, and regulatory requirements. Discover the country's public procurement policies and economic background.


Analyzing Mexico’s Pharmaceutical Landscape: Challenges and Opportunities
E N D
Presentation Transcript
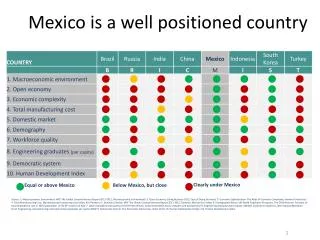
Country Presentations Mexico
Mexico’s Pharmaceutical DNA and Key Challenges and Opportunities
Mexico’s Pharmaceutical DNA • Health system • Population aged 65 and over predicted to reach 21% of total by 2050 • Public expenditure on health lowest in OECD (as of 2010) • Annual growth rates of health expenditure below OECD average • Pharmaceutical Environment • Pharmaceutical expenditure represents high percentage of total health expenditure • Private out-of-pocket (OOP) spending represents 95% of spending on pharmaceuticals • Pricing and Reimbursement (P&R) • Price caps on patented medicines based on international referencing • Reimbursement limited mainly to generic medicines, based on health technology assessment (HTA) Intellectual Property (IP) • Biopharmaceutical IP protection fairly standard, except limitations in ability to enforce patents • Counterfeited medicines problematic, particularly in private pharmacies
Presentation Overview • Demographic background • Macro-economic background • Health system overview • Pharmaceutical overview • Pricing and reimbursement policies • Intellectual property environment • Biopharmaceutical regulatory requirements • Public procurement policies – pharmaceuticals
Demographics • Total population: 34 million • Annual population growth rates: between 0.79% and 1.36% 2000-2010 • Dependency population 2010 • Youth population (aged less than 15): 28.1% • Elderly population (aged 65 and over): 5.9% • Dependency population aged 65 and over growing rapidly: predicted to reach 21.2% of total population by 2050
Dependency Population, Trend and Forecast, 2000-2050 Source: OECD Stat (2012)
Macroeconomic Background • GDP per head: USD 15,195 (2010) • OECD average USD 33,976 (2010) • Annual GDP growth 2011: 4.5% • OECD average 2011: 1.8% • Volatile economic growth rates over past decade – greater highs and lows than US, EU27 and OECD averages • Low national debt: 27% of GDP in 2010 • OECD central government debt average close to 80%
Real GDP % Growth 1997-2010 Source: OECD Stat (2012)
General Health System Facts • Several vertically integrated public insurers/providers for different parts of population (social security, low-income, unemployed/self-employed) • Universal coverage (through expansion of public insurance schemes) targeted in 2012 • Outside social security system, financing mix of MoH and states • Actual coverage and medical services provided by social security system and State Health Services • Out-of-pocket spending highest in the OECD (49% in 2010) • Health policy fragmented, divided between federal and state governments • Large private sector (represents 50% of health spending)
Health Expenditure • TEH 6.2% of GDP in 2010 (latest figures) • 3.3% lower than OECD average of 9.5% (2010) • 47.3% of total health spending in 2010 public expenditure • OECD average 72% (2010) • 3-4% increase in public expenditure as % of TEH since 2005 • Annual average growth rate (AAGR) total health expenditure 3.8% per year 2000-2009 • OECD average 4.3% • AAGR public sector 4.0% per year 2000-2009 • OECD average 4.5% • Out of pocket spending as % of TEH 49% 2010 • OECD average 19.5 % (2010)
Total Expenditure on Health % of GDP, 1990-2010 Source: OECD Stat (2012)
Public Expenditure on Health, % TEH, Mexico, 1990-2011 Source: OECD Stat (2012)
AAGR Mexico and OECD Averages, 2000-2009 Source: OECD Stat (2012)
Public expenditure and out-of-pocket payments, % total expenditure on health, 2010 Source: OECD Stat (2012)
Pharmaceutical Overview (1) • Pharmaceuticals partially covered through various health insurance schemes • Public insurance provision dominated by generics • Traditionally, three main classes of medicines in the market: • Innovative (patented); • “Interchangeable generics” (approved and registered by MoH based on bioequivalence tests); and • “Similar generics” or similares (generics approved and registered without bioequivalence tests) • Public insurance provision of similaresphased out 2005-present
Pharmaceutical Overview (2) • High volume of medicines sold through private pharmacies due to insufficient public health insurance coverage of pharmaceuticals • Private financing of medicines far outweighs public financing: 80% vs. 20% • Majority of private spending on medicines is OOP (over 95%) • Still, overall, lowest consumption of pharmaceutical products per capita in OECD
Snapshot of Pharmaceutical Spending • Pharmaceutical spending as % TEH 2nd highest in OECD (27.1% in 2009) • Per capita spending 2nd lowest in OECD (2009) • Medium growth in per capita pharmaceutical spending in 2000s, from very low rates at beginning of decade • Between 1999-2009 per capita spending almost tripled from USD 87.3 to USD 249.9 (PPP) • Incremental growth expected: pharmaceutical sales projected to rise from US$13 billion (MXN161 billion) in 2011 to US$18 billion in 2015
Total Expenditure on Pharmaceuticals and Other Medical Non-durables, % TEH, Mexico, 1999-2009 Source: OECD Stat (2012)
Total Expenditure on Pharmaceuticals, % of TEH, 1999-2009, Mexico and Select OECD Countries Source: OECD Stat (2012)
Total Per Capita Spending, Pharmaceuticals and Other Medical Non-durables, US$ PPP, Mexico, 1999-2009 Source: OECD Stat (2012)
Total Per Capita Spending, Pharmaceuticals and Other Medical non-durables, US$ PPP, 2010 or Closest Year, OECD Source: OECD Stat (2012)
Pricing Policies Patented Medicines • Maximum retail prices capped by Ministry of Economy (mainly for private sector) • Use international reference pricing • Calculated on basis of the average ex-factory price of the previous quarter in the six largest markets for a given product globally • Governed by General Law on Health, Article 31 • Lack of across-the-board compliance by pharmacies, poorly regulated • Little competition in public sector prices for patented medicines (due to centralized purchasing price)
Generic Pricing Policies • Pricing • Public sector: no central price control, wide variation in prices by state or insurance scheme • Private sector: generics/off-patent medicines no price control • Policies Promoting the Use of Generics • Federal Commission for the Protection against Sanitary Risks (COFEPRIS) creates Interchangeable Generics List • Listed by INN, includes both generic and original products • Public sector insurance schemes use list as well as National Formulary to encourage (but not require) generic prescription • Generic consumption through public insurance coverage still limited
Reimbursement Policies • All public institutions and insurance schemes governed by National Formulary (CuadroBásico y Catálogo de Medicamientos) • Set by National Formulary Committee (CICBISS) of the General Health Council (CSG) • Dictates first, second and third lines of treatment • 4-5 months for review and decision • Listed by INN, each assigned a code • Limits medicines available to 932 INNs (2011) based on market authorization and Economic Evaluation Study (EEE) • Large majority are off-patent • Published annually
HTA Policies • Economic Evaluation Study (EEE) compares current standard treatment with product proposed for inclusion in CuadroBásico • Mandated by CICBISS Internal Regulation, Article 24 • Submitted by company, CICBISS reviews according to CSG guidelines • Must include at least 1 of the following comparisons: • Cost-minimization – cost savings compared to equally effective comparators • Cost-effectiveness – incremental cost-effectiveness ratio relative to GDP per capita per additional years of life • Cost-utility – same as above, using QALY as denominator • Cost-benefit – rate of return relative to public debt issued by Federal Government
Pharmaceutical IP Overview • Biopharmaceutical IP protection fairly standard, but not fully in line with international standards and bilateral commitments • Biotech inventions patentable, except in natural state or processes of reproduction • Patent linkage system ineffective, lacks transparency • IP enforcement improving but insufficient • Significant delays in prosecution common • Injunctions frequently ineffective • Sales of counterfeit medicines in pharmacies undeterred, although efforts to contain are increasingly routine
Pharmaceutical ‘Patent Linkage’ Mechanism • Basic linkage system exists based on Presidential Decree (2003) • Requires generic applicants to comment on the patent status of the reference product • Potential infringement issues resolved by Mexican Patent Office (IMPI) and COFEPRIS and published by IMPI • Significant weaknesses: • Does not involve notification or consultation of the patent holder • Only applies to substance patents; despite 2008 Supreme Court ruling requiring inclusion of formulation and use patents, not consistently applied • Process often delayed and ineffective
PTE and RDP • Mexico does not offer patent term extensions for pharmaceutical products • COFEPRIS introduced 5 year regulatory data protection term in 2012 • RDP only applies to NCEs thus far • Implementation of RDP still uncertain
Marketing Authorization for Chemical Entities • MoH Federal Commission for the Protection against Health Risks (COFEPRIS) responsible for drug testing and approval • New drug submissions required to undergo safety, efficacy and quality testing • All generics now required to submit bioequivalence tests • Re-registration required every 5 years (elimination of similares) • COFEPRIS actively monitors availability of unapproved/sub-standard drugs • Recent delays in approvals; 2011-12 new target maximum approval time 180 days • COFEPRIS observes and has adopted guidelines of International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH)
Removal of Similares from the Mexican Market • Similaresformalized in 1998 reform to General Law on Health • Reform identified three classes of drugs approved by MoH (innovative, interchangeable generics and similar generics) • Similaresdid not undergo bioequivalence tests; not sanctioned as interchangeable with the original drug • Accounted for 36% of medicines sold in private sector in 2002 • Quality and safety debate over non-bioequivalence tested products in both public and private sectors 2002-2005 • Effort to phase out similares from the market, 2005-2010 • 2005 amendment to General Law on Health, Art.376 required all generics to undergo bioequivalence tests • All generics registered prior to 2005 had to be re-registered using bioequivalence tests by 2010 • Not yet fully implemented; severe delays in re-registration (1,000 applications remaining in Oct 2012)
Marketing Authorization for Biologics and Biosimilars • Biological drugs undergo the same procedure and testing requirements as new chemical entities • “Biocomparables” (biosimilars) pathway came into force in 2012 (Mexican Health Law, Art 222bis and COFEPRIS guidelines) • Mexican biosimilar pathway de facto resembles that used by the European Medicines Agency • Biologic similares(bio-similares) being phased out along with other similares • RDP protection/periods do not exist for biologics and biosimilars
Public Procurement Policies – Pharmaceuticals (1) • General characteristics of the procurement system • Relies on INN (as per the CuadroBásico) • Favors generics over innovative products • Decisions based largely on price • Value plays a role mainly to the extent it is a factor of a product’s inclusion in the CuadroBásico
Public Procurement Policies – Pharmaceuticals (2) • Phase I: Setting of centralized procurement price • Coordinating Commission for Negotiating the Price of Medicines and Other Health Inputs (CCNPMIS) annually secures single price for given product for all public institutions • Composed of representatives of MoH, MoEcon, public health insurers • Principle objective: uniform price reductions • Phase II: Actual procurement by individual institutions • Based on centrally agreed price • Mandatory to tender for products included in CuadroBásico(generics where possible) • States handle purchasing of non-essential medicines for all MoH-dependent schemes • Wide variation in average drug prices among states