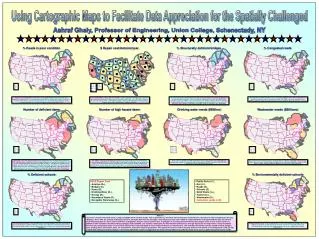
Elonva in poor responders
390 likes | 1.2k Vues
Elonva in poor responders. Shahar Kol August 2014. Who is a poor responder?. Human Reproduction 2011. Etiology?. Depletion of ovarian follicle pool Insufficient initial follicle number Accelerated loss Ovarian follicle dysfunction Signaling defect Enzyme deficiency Autoimmunity.

Elonva in poor responders
E N D
Presentation Transcript
Elonva in poor responders ShaharKol August 2014
Who is a poor responder? Human Reproduction 2011
Etiology? • Depletion of ovarian follicle pool • Insufficient initial follicle number • Accelerated loss • Ovarian follicle dysfunction • Signaling defect • Enzyme deficiency • Autoimmunity
Risk factors • Advanced maternal age • Genetic conditions • Turner, FMR1, X deletions • Gene mutation: FSHR, LHR • Acquired conditions • Endomertioma • Chemo/radiotherapy • Ovarian surgery
Prediction of poor ovarian response (POR) Broer et al, 2013
Which protocol? Survey on POR from 196 centers in 45 countries, 124,700 cycles
Treatment Protocols for poor responders • “There is insufficient evidence to support the routine use of any particular intervention either for pituitary down regulation, ovarian stimulation or adjuvant therapy in the management of poor responders to controlled ovarian stimulation in IVF”. 2010
Adjuvant therapy • Androgens (DHEA, testosterone, LH) • Growth hormone • Co-enzyme Q10 supplementation • Other?
Significance of POR • Poor prognosis for IVF success • Increased miscarriage risk • Early menopause
Elonva in the older Age group • Primary objective • To examine the efficacy and safety of a single injection of corifollitropin alpha vs daily recombinant FSH (rFSH) for controlled ovarian stimulation in women aged 35-42 years
Objective: • To identify whether women with poor ovarian response may benefit from treatment with corifollitropinalfa in a GnRHantagonist protocol. • Design: Retrospective pilot study. • Intervention: Corifollitropinalfa (150 mg) followed by 300 IU rFSH in a GnRH antagonist protocol. • Comparative cohort: short agonist, hMG 300-450 IU/d Polyzos et al. FertilSteril 2013
Conclusion: • Treatment of poor ovarian responders, as described by the Bologna criteria, with corifollitropinalfa in a GnRH antagonist protocol results in low pregnancy rates, similarly to conventional stimulation with a short agonist protocol. Polyzos et al. FertilSteril 2013
Will sequential administration of highly purified (hp)-HMG after corifollitropin alfa in a GnRH antagonist protocol benefit women with poor ovarian response according to the Bologna criteria? • Retrospective pilot study. Polyzos et al, 2013
Endocrine profiles during the follicular phase in women who are poor ovarian responders, according to age E2, estradiol. *P . 0.05 for all comparisons between age groups at Days 2, 7, 9 and day of hCG triggering.
Conclusion • Corifollitropinalfa followed by hp-HMG in a GnRH antagonist protocol results in very promising pregnancy rates in young (<40 years old) poor ovarian responders fulfilling the Bologna criteria.
Results in POR by age • Retrospective study • 485 patients, 823 cycles • 201<40 years, 284>40. • Gonadotropin daily dose ≥ 300 IU (FSH and/or hMG). Polyzos et al , 2014
Follicular recruitment is a random event • Recruitment occurs all the time. • This explains our ability to start stimulation in luteal phase. • The number of recruitable follicles in any given time point changes by chance. • The specific type of gonadotropins plays a secondary role.
Potential advantage of Elonva • In the natural follicular phase FSH decreases until the midcycle surge.
Flare effect • Without using GnRH agonist • No cysts formation, no LH rise • Robust recruitment of all available responsive follicles?
Does the different pharmacokinetic Profile of corifollitropinalfaresult in a significantly higher number of oocytes retrieved compared with rFSH? Engage Study, Devroey et al , 2009
Elonva: reducing Treatment burden • POR patients are prone to have repeated IVF trials. • Reduced complexity and treatment burden • Sort treatment cycle (antagonist-based) • Fewer overall injections • Fewer injections per day • Fewer drop-out patients.
In conclusion • Elonva is an important addition to our fertility drugs arsenal. • the advantage of Elonva in the treatment of POR is yet to be defined by randomized controlled studies, and by personal experience by each treating physician in the field of ART. Thank you