Ozone Hole
Ozone Hole. History of the Ozone Discovery. Dramatic loss of ozone in the lower stratosphere over Antarctica was first noticed in the 1970s by a research group from the British Antarctic Survey (BAS) who were monitoring the atmosphere above Antarctica. What is the ozone hole?.

Ozone Hole
E N D
Presentation Transcript
Ozone Hole www.assignmentpoint.com
History of the Ozone Discovery • Dramatic loss of ozone in the lower stratosphere over Antarctica was first noticed in the 1970s by a research group from the British Antarctic Survey (BAS) who were monitoring the atmosphere above Antarctica www.assignmentpoint.com
What is the ozone hole? • News media confuses it with the problem of global warming • ozone contributes to the greenhouse effect • over Antarctica (and the Arctic), stratospheric ozone depleted over past 15 years at certain times of the year • hole presently size Antarctica, 10km altitude - lower stratosphere www.assignmentpoint.com
What is ozone? • Ozone forms a layer in the stratosphere, thinnest in the tropics (around the equator) and denser towards the poles • measured in Dobson units (DU) • ~260 DU near the tropics www.assignmentpoint.com
What is a Dobson unit? • 1 Dobson Unit (DU) is defined to be 0.01 mm thickness at STP - (0C and 1 atmos press). • A slab 3mm thick corresponds to 300 DU www.assignmentpoint.com
How is ozone formed? UV radiation strikes the O2 molecule and splits it, atomic oxygen associates itself with another O2 molecule – simplistic version www.assignmentpoint.com
Climatology of ozone Mean climatology Annual cycle www.assignmentpoint.com
Synoptic variability of ozone Ozone also has substantial variability on smaller time scale www.assignmentpoint.com
Vertical variation of ozone www.assignmentpoint.com
Source, sink and reservoirs Ozone is in a fluid state of creation and destruction www.assignmentpoint.com
How ironic . . . • at ground level, ozone is a health hazard • major constituent of photochemical smog • in the stratosphere, it absorbs potentially harmful ultra-violet (UV – 240-320nm harmful) radiation • Protects from skin cancer, etc www.assignmentpoint.com
“Chapman Reactions” • Ozone is formed by: O2 + hv -> O + O (1) • Ozone can reform resulting in no net loss of ozone: O3 + hv -> O2 + O (3) O + O2 -> O3 (2) • Ozone is also destroyed by the following reaction: O + O3 -> O2 + O2 (4) www.assignmentpoint.com
Comparison of reactions • Reaction (2) slower with increasing altitude • Reaction (3) faster with increasing altitude • Lower in stratosphere, atmosphere denser, UV absorption increases – ozone peaks 20km • Closer to surface, UV level decr, ozone decr www.assignmentpoint.com
Chemical processes ozone depletion • Chlorine, bromine – from human activities • Chlorine carriers – hydrochloric acid, chlorine nitrate • Other impt cpds – nitric acid, dinitrogen pentoxide • Right conditions necessary for chemical reactions to occur • On surface of PSC • Denoxification – slows removal ClO www.assignmentpoint.com
Not there yet - • We still have many more atoms ozone than active chlorine • How do we destroy all the ozone?? www.assignmentpoint.com
One more step: • Only have molecular chlorine (Cl2) • Require atomic chlorine to destroy ozone • Via “photodissociation” • Cl2 + hv -> Cl + Cl • Key to timing of ozone hole • Finally – catalytic destruction of ozone www.assignmentpoint.com
Final stage • Catalytic cycle – molecules significantly changes or enables a reaction cycle without being altered by the cycle itself www.assignmentpoint.com
Ozone loss recipe - summary • Polar winter polar vortex isolates air within • Cold temperatures Polar Stratospheric Clouds vortex air isolated cold temperatures & PSC’s persist • Heterogeneous reactions allow reservoir species of chlorine & bromine - rapidly converted to more active forms. • No ozone loss until sunlight returns production active chlorine initiates catalytic ozone destruction ozone loss rapid www.assignmentpoint.com
The Antarctic polar vortex www.assignmentpoint.com
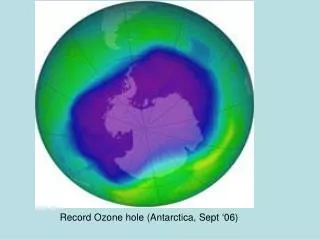
Ozone loss over Antarctica • most dramatic in the lower stratosphere • nearly all the ozone depleted • area the size of Antarctica • many km thick • most pronounced in spring/October • persists two months • December – moves Falklands, S Georgia, S Am www.assignmentpoint.com
Evolution ClONO2 – North Pole – winter 1994 • http://www.atm.ch.cam.ac.uk/tour/tour_mpeg/anim_clono2.mpg • Winter – no light – ClONO2 destroyed – sunlight returns – ClONO2 recovery – edge vortex – doughnut shape – ‘chlorine nitrate collar’ www.assignmentpoint.com
What causes the depletion? • release of manmade chemicals – • CFC - refrigerants, aerosol sprays, solvents and foam-blowing agents • halogen compounds - Fire fighters used bromine-containing halogens to put out fires • NOx www.assignmentpoint.com
One chlorine atom - average - destroys one thousand ozone molecules before converted into form harmless to ozone www.assignmentpoint.com
Atmospheric Chlorine cycle www.assignmentpoint.com
Vertical Structure of Ozone Cycle www.assignmentpoint.com
CFCs and Ozone depletion Reservoirs Reactions www.assignmentpoint.com
Nitrous oxide cycle www.assignmentpoint.com
Ozone depletion through nitrous oxide www.assignmentpoint.com
Sources that harm ozone layer www.assignmentpoint.com
Sources of chloroflurocarbons www.assignmentpoint.com
Total ozone –October monthly averages Halley Bay, Antarctica www.assignmentpoint.com
Monthly averages for October www.assignmentpoint.com
TOMS Satellite Measurements • Total Ozone Mapping Spectrometer • Based on backscattered light • UV range • Dobson units (DU) www.assignmentpoint.com
Ozone Hole – Sept 22-Oct 6 2002 & 2003 www.assignmentpoint.com
Ozone hole size • 1.5 x USA • Australia 8,923,000 • USA 9,363,130 • Europe 10,498,000 • Antarctica 13,340,000 • Russia 17,078,000 • N America 25,349,000 • Africa 30,355,000 sq km www.assignmentpoint.com
Need for a cold dark place….. www.assignmentpoint.com
What is being done? • First global agreement - restrict CFCs - Montreal Protocol - 1987 • European Community countries have even stricter measures • Was anticipated - recovery of the ozone layer within 50 years of 2000 World Meteorological Organisation (WMO reports #25, #37) www.assignmentpoint.com
Is the ozone loss only in Antarctica? • Arctic – ‘low ozone event’ • S America – from Antarctica hole • Volcanic ozone hole • European ozone hole - heavily populated northern mid-latitudes (30-60N) – formation smaller, much slower www.assignmentpoint.com
Why is the loss more dramatic at the poles? • Polar meteorology • Polar vortex – winter polar night • Polar stratospheric clouds (-80C) • nitric acid trihydrate • Chemical reactions • occur on surface PSC’s • Occur very fast www.assignmentpoint.com
What drives the vortex? www.assignmentpoint.com
Vortex explanation • mid-May - onset SH winter • Antarctic stratosphere cools • descends closer to the surface • Coriolis effect - sets up strong westerly circulation (@ SP) • forms oblong vortex • temperatures - lower stratosphere cool < -80C PCS’s www.assignmentpoint.com
South American Ozone • The Ozone hole has reached Argentina, Chile and The Falkland Islands since the 1990's. • Ozone levels dropped down 70% in some areas. • The protective level of ozone dropped below 150 DU in some areas. www.assignmentpoint.com
Arctic ozone hole? www.assignmentpoint.com
What does this look like on population density map? www.assignmentpoint.com
Any satellite evidence? May 25, 2000 – “low ozone event” www.assignmentpoint.com
What do these clouds look like? www.assignmentpoint.com
Implications of ‘low ozone event’ • Could be blown south by high-altitude winds across heavily populated regions • 10% reduction ozone layer 25% imcr non-melanoma skin cancer – temperate climates by 2050 • Arctic ozone recovery may not be as quick as Antarctic www.assignmentpoint.com
Why the different response? • Polar stratospheric clouds surfaces where benign forms of chlorine reactive ozone-destroying forms • Remove nitrogen compounds that moderate destructive impact of chlorine - Dr. Phil De Cola, Atmospheric Chemistry Program Manager, NASA Headquarters, Washington, DC. • Presently – nitric acid stays longer in Arctic – reduces amount reactive chlorine www.assignmentpoint.com