Ozone Hole
The Earth's atmosphere, though incredibly thin at just 120 km compared to the planet's size, plays a crucial role in sustaining life by blocking harmful UV radiation through the ozone layer. This layer has suffered significant depletion due to chlorofluorocarbons (CFCs), leading to the infamous ozone hole. The consequences are dire, including increased skin cancer rates and adverse effects on agriculture and marine ecosystems. International efforts like the Montreal Protocol aim to phase out CFCs and restore the ozone layer, highlighting the need for collective action to protect our planet.

Ozone Hole
E N D
Presentation Transcript
Earth’s Atmosphere Compared to the size of the Earth (12000 km) The atmosphere is very thin (120 km) http://www.gsfc.nasa.gov/gsfc/earth/pinatuboimages.htm
If theEarthis compared to this Orange the Earth’s atmosphere would be thinner than the layer of pesticide on this Orange’s surface
Two Atmosphere Layers • Stratosphere is above Troposphere • Ozone Layer blocks UV radiation • Troposphere is where we live • Weather occurs here • 72% of all air is below the cruising altitude of commercial airliners (33000 ft)
Air Pollution • Ozone Pollution vs. the Ozone Hole
Ozone – Two Faces • Troposphere – Bad Ozone • Pollution • Photochemical smog • Eye irritant • Stratosphere – Good Ozone • Blocks UV radiation • Hole caused by depletion due to CFC’s
Harmful effects of UV radiation • Skin cancer such as melanoma(ultraviolet radiation can destroy acids in DNA) • Cataracts and sun burning • Suppression of immune systems • Adverse impact on crops and animals • Reduction in the growth of ocean phytoplankton • Degradation of paints and plastic material
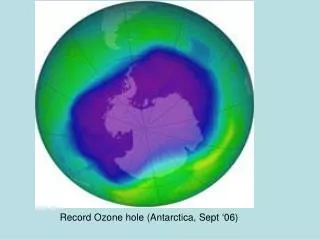
Destroying Good Ozone • Chloroflourocarbons were first created in 1928 as non-toxic, non-flamable refrigerants, and were first produced commercially in the 1930's by DuPont • In 1974, a laboratory study demonstrated the ability of CFC's to catalytically breakdown Ozone in the presence of high frequency UV light • Cl + O3 -> ClO + O2 ClO + O3 ---> 2 O2 + Cl • In a 1985, a study summarized data that had been collected by the British Antartic Survey showing that ozone levels had dropped to 10% below normal January levels for Antarctica. • http://www.nas.nasa.gov/About/Education/Ozone/history.html
Ultraviolet light hits a chlorofluorocarbon (CFC) molecule, such as CFCl3, breaking off a chlorine atom and leaving CFCl2. Sun Cl Cl Once free, the chlorine atom is off to attack another ozone molecule and begin the cycle again. C Cl F UV radiation Cl Cl O O A free oxygen atom pulls the oxygen atom off the chlorine monoxide molecule to form O2. The chlorine atom attacks an ozone (O3) molecule, pulling an oxygen atom off it and leaving an oxygen molecule (O2). Cl Cl O O O O O The chlorine atom and the oxygen atom join to form a chlorine monoxide molecule (ClO) Cl O O O
A single chlorine atom removes about 100,000 ozone molecules before it is taken out of operation by other substances
Montreal Protocol • An international treaty designed to protect the ozone layer • phasing out production of number of substances believed to be responsible for ozone depletion • Effective January 1, 1989 • Five revisions • 1990 (London) • 1992 (Copenhagen) • 1995 (Vienna) • 1997 (Montreal) • 1999 (Beijing)