Thin Film Growth
290 likes | 1.3k Vues
Thin Film Growth. Lecture 12 G.J. Mankey gmankey@mint.ua.edu. Factors Influencing Film Growth. Surface energy. Interfacial energy. Substrate temperature. Interface crystallography. Type of deposition source. Background gas. Surfactants.

Thin Film Growth
E N D
Presentation Transcript
Thin Film Growth Lecture 12 G.J. Mankey gmankey@mint.ua.edu
Factors Influencing Film Growth • Surface energy. • Interfacial energy. • Substrate temperature. • Interface crystallography. • Type of deposition source. • Background gas. • Surfactants.
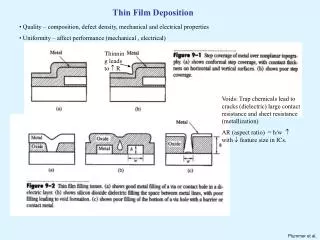
Bauer (1980s) described film growth in terms of surface energy with the relation: It states that for layer by layer growth to occur, the surface energy of the growing film must be less than or equal to the surface energy of the substrate minus the interfacial energy of the film and substrate. The equation is based on the assumption that the substrate temperature is high enough to achieve thermodynamic equilibrium. Surface Energy
Equilibrium Growth Modes • Layer-by layer or Frank-van der Merwe--the film grows with only the surface layer having fractional coverage at any instant i.e. the "interface width" at the film vacuum interface is one atomic layer. • Island or Volmer-Weber--the film does not "wet" the substrate since the Bauer condition is not met. The "interface width" of the growing surface increases with time. • Layer+island or Stranski-Kastranov--After wetting the substrate, the film grows in an island mode and the interface width increases with time.
Metastable Growth Modes • Diffusion-limited--the incoming atoms "stick where they land" and the film growth is characterized by a interface width which increases as da where a is the power law exponent (1/4 for perfect diffusion-limited growth). • Downward funneling--The impinging atoms maintain a component of vertical momentum and tend to fill in the lower areas of the interface, so the interface width saturates at 3-7 atomic layers.
Interfacial Energy • Some materials have a negative enthalpy of mixing with one another--they are miscible. • These materials tend to mix strongly at the film substrate interface to give a film-substrate "interface width" that may extend over many atomic layers. • One example is transition metals on Si which tend to form silicides with varying stoichiometry over the vertical interface width.
Immiscible Materials • An immiscible film-substrate combination will generally form an atomically-sharp interface, however there are some examples are in the literature where the epitaxial strain at the interface will change the situation. • If the Bauer condition is satisfied, layer or layer+island growth will be observed. • When the film has a high surface energy compared to the substrate, a layer of substrate material may segregate to the surface with the film sandwiched in the middle.
Substrate Temperature • The substrate temperature and flux are the primary factors that determine the resulting film morphology. • Low substrate temperatures or high flux rates (>1 nm/sec) result in metastable thermodynamic phases. • Interface mixing and substrate segregation may be suppressed for low temperature growth. • Details depend on materials properties such as melting points, epitaxial mismatch and enthalpy of mixing. • Atomically smooth films are usually the exception rather than the rule.
Interface Crystallography • If the growing film crystal structure has a specific relation to the substrate crystallography the film is epitaxial which means the film and substrate have an in-plane orientation relationship. • If the film has the same crystal structure as well as the same lattice constant then the film is pseudomorphic to the substrate. • If only an out of plane relationship is maintained then the film is highly textured with most of the surface comprised of the same crystallographic plane and rotated domains in the plane of the film.
Deposition Sources • Evaporators--the incoming atoms have kinetic energies of the order of less than 1 eV--the atoms have a small amount of energy to dissipate on the growing surface. • Sputter sources--the incoming atoms may have energies as high as several hundred eV--the incoming atoms are likely to mix strongly at the interface with the film since their energies are sufficient to break bonds in most solids. • Pulsed laser deposition--kinetic energies similar to evaporators with the incoming atoms often in highly excited electronic states or multiply ionized states.
Background Gas • In poor vacuum (>10-7 mbar) the gas is mostly water vapor. The water adsorbed on the surface may increase the mobility of surface atoms and the oxygen may be incorporated in the growing film. • Intermediate vacuum (10-9 mbar < P < 10-7 mbar) the presence of CO and CO2 on the surface may dominate the growth process--Look for C to be incorporated in the film. • The gas may help to stabilize metastable phases--see vast literature of Fe/Cu(100) system.
Surfactants • A surfactant bonds more strongly to the film material than the substrate, so the Volmer-Weber growth mode may be suppressed. • Surfactants are characterized by a "floating out" efficiency or capability of the surfactant atoms to remain on the surface without being incorporated in the film. • Surfactants may also significantly alter surface morphology to enable the formation of atomically smooth films. • It is difficult to remove surfactants after they have done their job.