Thin Film
Thin Film . By S h a d i a M. S .E l a y y a t. Out Line . Objective Review Type of materials Type of semiconductor P-N junction Thin film Definition Type Thin film deposition Application References . Objectives The main objectives of this research are:-


Thin Film
E N D
Presentation Transcript
Thin Film By ShadiaM.S.Elayyat
Out Line • Objective • Review • Type of materials • Type of semiconductor • P-N junction • Thin film • Definition • Type • Thin film deposition • Application • References
Objectives • The main objectives of this research are:- • Study the characteristics of the materials • Study P-N junction • Discus and study the thin film deposition • Study the application and uses of thin film
Materials Conductor Semiconductor Insulator plastics, paper, glass gold, silver, nickel, iron. Silicon , Germanium
Conductor • mostly metals • have the highest conductivities • The conductivity decrease as the temperature increase
Insulator • have high resistivity • does not conduct an electric current under the influence of an electric field • Example : • Plastics • Paper • glass
semiconductor Semiconductors are materials whose electrical properties lie between Conductors and Insulators. Some common semiconductors elemental • Si - Silicon (most common) • Ge - Germanium compound • GaAs - Gallium arsenide • GaP - Gallium phosphide • AlAs - Aluminum arsenide • AlP - Aluminum phosphide • InP - Indium Phosphide
The resistivity of the semiconductor materials decrease as the temperature increase as shown if the figure
Valance Band The outermost orbital of an atom, where electrons are so tightly bounded that, they can not be removed as free electron Conduction Band This is the highest energy level or orbital in outer most shell, in which electrons are free enough to move. Band Gap There is one energy gap separates these two bands, - the valance band and conduction band. This gap is called forbidden energy gap Metal very small energy gab Semiconductor moderate E.G Insulator Large energy gab
insulators The electrical conductivity at room temperature is quite different for each of these three kinds of materials Metals have the highest conductivities followed by semiconductors and then by insulators
Type of semiconductor Two type of semiconductor • Intrinsic(undoped ) semiconductor: the number of excited electrons and the number of holes are equal: n = p
Extrinsic semiconductor : is a semiconductor that has been doped. • Doping involves adding dopant atoms to an intrinsic semiconductor, which changes the electron and hole carrier concentrations of the semiconductor at thermal equilibrium. • an extrinsic semiconductor classify it as either an n-type or p-type semiconductor
P-type semiconductor is an intrinsic semiconductor in which an impurity acting as an acceptor(number of holes grater than number of electrons ) • N-type semiconductor is an intrinsic semiconductor in which a donor impurity has been intentionally introduced(number of electrons much grater than holes).
The impurity could be indium or gallium, both of which have only three valence electrons. • Majority carriers are holes • Minority carriers are • electrons Lattice structure of p-type semiconductor
The impurity could be arsenic ,which have only five valence electrons. • Majority carriers are electrons • Minority carriers are • holes Lattice structure of N-type semiconductor
P-N Junction • A p–n junction is a boundary or interface between two types of semiconductor material, P-type and N-type . • p–n junctions are building blocks of most semiconductor electronic devices such as diodes, transistors, solar cells, LEDs.
When the p-type and n-type semiconductors are join , electrons from the n region near the p–n interface diffuse to the p-type region. As electrons diffuse, they leave positively charged ions ,in the n region. The holes from the p-type region near the p–n interface diffuse to the n-type region, leaving fixed ions with negative charge. The regions nearby the p–n interfaces lose their neutrality , forming the depletion zone .
Forward bias • the p-type is connected with the positive terminal and the n-type is connected with the negative terminal • the holes in the P-type region and the electrons in the N-type region are pushed toward the junction. This reduces the width of the depletion zone.
Reverse bias • the p-type is connected with the negative terminal and the n-type is connected with the positive terminal • the voltage at the cathode is higher than that at the anode. So, no current will flow • the holes in the P-type material are pulled away from the junction, causing the width of the depletion zone to increase. Likewise, the electrons in the n-type material will also be pulled away from the junction.
Threshold voltage Break down voltage The reverse saturation current
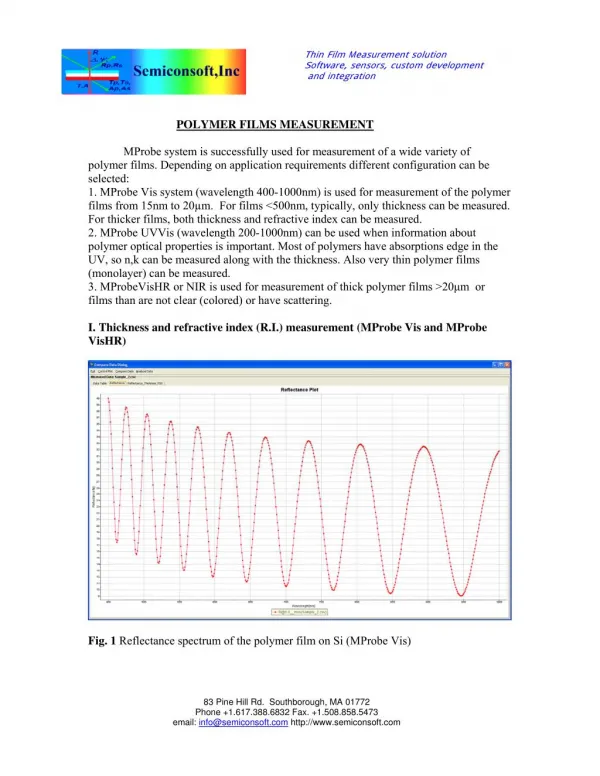
What is a "thin film" ? • thin = less than about one micron ( 10,000 Angstroms, 1000 nm) • film = crystalline or amorphous layer of material on a substrate
Thin films are crystalline or amorphous layers, typically 1 nm – 10 µm thick, deposited on a substrate.
Classification of Thin Films • single crystals • Polycrystalline • amorphous
Properties of Thin Films • not fully dense • under stress • quasi - two dimensional (very thin films)
Thin film Substrate(transparent conducting oxide ) glass
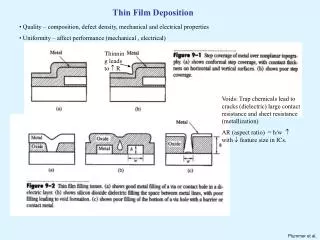
thin film deposition • Chemical Vapor Deposition • Physical Vapor Deposition • Electrodeposition • Langmuir-Blodgett
Chemical Vapor Deposition • process used to produce high-purity, high-performance solid materials • Reactive gases interact with substrate • Used to deposit Si and dielectrics Type of CVD • Atmospheric pressure – APCVD • Low pressure – LPCVD • Plasma enhanced – PECVD • High density plasma - HDPCVD
Chemical Vapor Deposition • Involves one or more gas phase species which react on a solid surface (substrate) to deposit a solid film. • the reaction is initiated by heating the substrate. Other mechanisms of supplying the activation energy necessary to initiate reactions include: laser CVD, photo CVD, and plasma enhanced CVD.
Physical Vapor Deposition There are two type of PVD :- • evaporation • Sputtering • Very few chemical reactions • deposits almost any material • Always performed in vacuum. • Vacuum increases mean free path of ions or atoms • Vacuum is typically less than 10–4Torr
A thermal evaporator uses an electric resistance heater to melt the material and raise its vapor pressure to a useful range. This is done in a high vacuum, both to allow the vapor to reach the substrate without reacting with or scattering against other gas-phase atoms in the chamber, and reduce the incorporation of impurities from the residual gas in the vacuum chamber.
Sputtering (sputtering mean a process which atoms are ejected from a solid target material due to bombardment of the target by energetic particles).This way is one of the most flexible deposition techniques. It is especially useful for compounds or mixtures, where different components would otherwise tend to evaporate at different rates. Uses a plasma (usually a noble gas, such as argon) to knock material from a "target" a few atoms at a time. The target can be kept at a relatively low temperature, since the process is not one of evaporation. Sputtering widely used in the optical media. The manufacturing of all formats of CD, DVD, and BD are basically done with the help of this technique. It is a fast technique and also it provides a good thickness control
Applications • microelectronics - electrical conductors, electrical barriers, diffusion barriers . • magnetic sensors - • gas sensors • optics - anti-reflection coatings • solar cell fabrication • photodiodes • switching devices
Uses • microelectronic devices • telecommunication devices • wear resistant coatings • optical coatings (windows,solar cells) • Sensors • catalysts
Reference • Karl W. Böer, “Survey of Semiconductor Physics Volume II: Barriers, Junctions, Surfaces, and Devices”, Springer,617-619 (1992). • Always Learning, “Electronic Devices and Circuits”, Pearson Education India,26-27 (2008). • PreetiMaheshwari, “Electronic Components and Processes”, New Age International,25( 2006). • R.S.Sedha, “A Textbook of Electronic Circuits”, S. Chand, 32-45(2008).
S. Mani Naidu, Naidu S. Mani,” Applied Physics”, Pearson Education India,38-45( 2009). • Simone Raoux, Matthias Wuttig, “Phase Change Materials: Science and Applications”, Springer, 102(2010). • Masanori Okuyama, Yoshihiro Ishibashi, “Ferroelectric Thin Films: Basic Properties and Device Physics for Memory Applications”, Springer, (2005). • Y.Paulean,“Chemical Physics of Thin Film Deposition Processes for Micro- And Nano-Technologies”, Springer,vii-4( 2002).
any solid object in which an orderly three-dimensional arrangement of the atoms, ions, or molecules is repeated throughout the entire volume Single crystal
materials are solids that are composed of many crystallites of varying size and orientation. The variation in direction can be random (called random texture) or directed. • all common metals and many ceramics are polycrystalline Polycrystalline
non-crystalline solid is a solid that lacks the long-range order characteristic of a crystal. amorphous