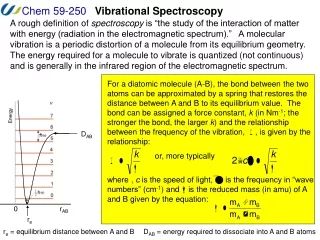
Vibrational spectroscopy
Vibrational spectroscopy. Chemical composition: finger print Bonding orientation: adsorption structure on surfaces . Infrared Spectroscopy (IR) High Resolution Electron Energy Loss Spectroscopy (HREELS) Surface Enhanced Raman Spectroscopy (SERS) Second Harmonic Generation (SHG)


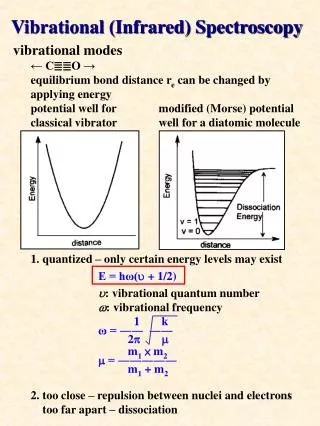
Vibrational spectroscopy
E N D
Presentation Transcript
Vibrational spectroscopy • Chemical composition: finger print • Bonding orientation: adsorption structure on surfaces Infrared Spectroscopy (IR) High Resolution Electron Energy Loss Spectroscopy (HREELS) Surface Enhanced Raman Spectroscopy (SERS) Second Harmonic Generation (SHG) Photo-acoustic Spectroscopy (PAS) Inelastic electron tunneling Spectroscopy (IETS) Inelastic Neutron Scattering (INS)
Surface Infrared spectroscopy • Refs: Y.J. Chabal, Surf. Sci. Rep. 8, 211 (1988) • F.M. Hoffman, Surf. Sci. Rep. 3, 107 (1983) • Transmission IR Spectroscopy • supported metal cataysts • IR transparent samples (Si) • Diffuse Reflectance Infrared Fourier Transform • Spectroscopy (DRIFTS) • refocus the diffusively scattered IR beam • high surface area catalytic samples • low surface area single crystals • Reflection-Absorption IR Spectroscopy ( RAIRS ) • specular reflected IR beam • single crystal samples • Multiple Internal Reflection Spectroscopy ( MIR ) or • Attenuated Total Reflection (ATR) • total internal reflection • SAM , polymer films
Background - I I0 + - • Transmission and absorption mode • Transmittance T = I/I0 = exp(kcl) • Absorbance A = ecl • k: absorption coefficient; e : absorptivity • c : concentration; l : cell thickness • Imaginary part of refractive index n = k • n = n + ik for absorbing medium • n = n for dielectric non-absorbing medium • needs to take reference and sample spectra • not popular for surface studies due to the large bulk contribution
Reflection The reflection angles Snell’s law n1/n2 = sinqi/sinqt Crtical angle: qc= sin-1(n2/n1) Intenstiy of the reflected light - Depend on polarizations Fresnel’s equations n = n + ik s-polarized light : || the plane of incidence Rs = [(n-secq)2+k2]/ [(n+secq)2+k2] p-polarized light : ㅗ the plane of incidence Rp = [(n-cosq)2+k2]/ [(n+cosq)2+k2] - qi must be large: grazing incidence for thin films on reflective surface the plane of incidence x Ep Es x qi qr qt
Phase shift , electric field, intensity of p-polarized light as a function of incidence angle from a metal surface s-pol: qㅗ n =3, k=30 Surface intensity function 180 0 Surface electric field E/E0 Phase shift on refelctions (E/Eo)2secq 60 40 p-pol :q|| 20 0 incidence angle 90 0 incidence angle 90 s-polarized light at the surface - uniform phase shift - vanishing E field at the surface p-polarized light at the surface - dependent on incidence angle - strong E field at large incidence angle, ie, grazing incidence Absorbance is proportional to E2 and area of surface as 1/cos q I ~ E2/cos q
Adsorbate covered surface Dielectric constant e = (n+ik)2 Ro R Vcauum e1n1 d Adsorbate e2(n2,k2) Ro R Metal e3(n3,k3) Absorption function A = (R- R0) /R0 = DR/R e3 >> e2~1, d<<l DRs/Rs = (8pdcosq/l)Im((e2– e3)/(1-e2)) DRp/Rp = (8pdcosq/ l)Im([(e2– e3)(1-(1/ e2e3)(e2+ e3)sin2q]/ (1-e2)(1/e3) )(1+ e3)sin2q] Reflectivity change of s-polarized light is negligibly small Assuming e3 >> e2 and cosq > e3-1 DRp/Rp = (8pdsin2q/lcosq)Im(-1/e2) a large reflectivity change at high incidence angle
Surface selection rule mfi = <f|m|i> 0, dm/dr 0 • The electric field of light and the molecule interact with surface electrons • The incident light must be p-polarized • Only vibrations with a dipole moment perpendicular to the surface • The incident light should be reflected at grazing incidence mM + mM + - - - + - mimage mimage + IR inactive IR active • for lying down molecules, molecular and image dipoles are cancelled out • for upright molecules, molecular and image dipoles are enhanced
Identification of adsorbate: high resolution : 2-4 cm-1 Orientation of adsorbed molecule by surface dipole selection rule How to confirm the metal-adsorbate bond ? - frequency shift of internal modes compared to gas-phase spectra - additional metal-molecule vibration: <800 cm-1 Frequency shift of internal and external modes for adsorbed layers - weakening of metal-molecule bond: n decreases as coordinationof surface atoms increases - formation of adsorbate islands - compression structures DR/R: 0.1~10-3 often small: sufficient for submonolayer sensitivity for molecule with strong dynamic dipole moment DR/R roughly linear with coverage, but not a good indicator of population Surface IR spectra of adsorbed molecules
Peak width and intensity homogeneous broadening - coupling to phonon - electron-hole creation inhomogeneous broadening - inhomogeneous distribution of harmonic oscillator - intermolecular interaction energy transport between molecule and surface dipole-surface interaction: dynamic dipole interaction
Instrumentation: RAIRS J.E. Reutt-Robey et al, JCP 93, 9113 (1990)
IR spectra of CO on Pd(100) • threefold:site : 1800~1900 cm-1 • bridge site: 1900~2000 cm-1 • on top site: 2000~2100 cm-1 Lower frequency shift compared to that of gas phase ? - Interaction with the vibrating dipole with the image dipole \ - Chemical effect due to backdonation, which change the CO bond strength Higher frequency shift as coverage - vibrational coupling : dipole-dipole, dipole-metal electrons - chemical effect: reduced backdonation into antibonding orbitals - electrostatic effect due to charge transfer between the metal and moelcule - intermolecular repulsion
IRRAS spectra CO on Pd(111)
High Resolution Electron Energy Loss Spectroscopy • Inelastic scattering of low energy electron beam • Energy loss due to the vibrational excitation • observe vib. modes parallel and perpendicular to the surface • Lower resolution 3meV (=24 cm-1 )(compare with IR ~2 cm-1) • Submonolayer sensitivity • can observe surface-atom vib. freq. <800 cm-1 Eo-E = hv I E Eo v + - Eo E
Dipole scattering Impact scattering Resonance scattering Scattering mechanism • Dipole scattering • electrons interact with the long range field at surface • electron momentum perpendicular to the surface normal is condserved • forward scattering by adsorbate • peaked in the specular position • elastic electrons: specular • inelastic electrons: near specular • vibration perpendicular to the surface normal can be excited • larger cross section for smaller Eo(~5 eV) E Eo g|| mM + mM g || + - - ki || ki kf - + - ㅗ mimage mimage ㅗ +
Impact scattering • short range interaction(~ a few A) of electron with atomic core potential • of surface • strong multiple scattering • Isotropic angular distributions • scattering probability depends on surface dipole amplitude and electron • energy • - favored by high incident electron energy > 50 eV • - off specular angle • lower scattering cross section the the dipole scattering Negative ion resonance scattering • short range interaction • electron trapped in empty Rydberg state of adsorbate • temporary negative ion • enhancement of vib. Intensity over relatively narrow range of Ei • very small cross- section off resonance • molecular orientation on surface
Examples: CO on W(100) 565 cm-1 ; W-C stretching 630 cm-1 : W-O stretching 363 cm-1 W-CO (on top) 2081 cm-1 CO stretching CO(g): 2140 cm-1
Interaction ions with solid • Charge transfer: neutralization of ion and electronic excitation • Kinetic energy transfer: sputtering, scattering e Auger neutralization Evac Resonance ionization EF Resonance neutralization Ei Quasi-resonance neutralization
Atomic and nuclear collision Impact parameter (b) scattering process energy transfer (Tc) ~1 A inelastic excitation 10eV of valence electrons ~10-1 A inelastic excitation 100eV of L-shell electrons ~10-2 A inelastic excitation 1 keV of K-shell electrons ~ 10-4 A elastic scattering ~100keV from nuclei
Ion scattering spectroscopy Low energy ion scattering (LEIS): 0.5 ~ 3 keV Medium energy ion scattering (MEIS): 10~500 keV High energy ion scattering (HEIS) or Rutherford backscattering spectroscopy (RBS): 0.5 ~5 MeV Binary elastic collision Kinematic factor K= E1/Eo E1/Eo = [((M22–M12)sinq)1/2+M1cosq) /(M2+M1)]2 M1,M2 : mass of incident atom and target q = scattering angle
Blocking, shadowing, and channeling effect • scattering cross section • at a certain angle depend • on atomic potentials of incident • and substrate atoms • scattering depend on incident • angle and impact parameter • lower ion energy, • larger shadow cone
Scattering cross section 2pbdb = s(q) 2psinqdq s(q) = b(db/d q)/sin q = # of scattered paricles into dW/total # of incident particles Rutherford formula ds /dW = [Z1 Z2e2/4Ecsinqc/2]2 Ec = [M2/(M1+M2)]Eo dq q b db
Quantitative analysis Total # of particles of impurity mas M3, atomic number Z3, surface density N3(atoms/cm2) The measured yield Y3 Y3 = N3 (ds /dW) DW Q Q: measured # of incident particles DW : solid angle accepted by detector - N3 can be determined typically with an accuracy better than 10%
Stopping power and depth resolution • the rate of energy loss dE/dx depends on mass of projectiles, traget, and • incident energy • for 0.5~2.0 MeV, dE/dx is independent of energy • Depth resolution: 30~100 Å Electronic stopping during going in Elastic scattering Electronic stopping during going out Final Energy of a particle at normal incidence E1 = Eo – DEin – Es - DEout
Sputtering yield:angle dependence • varies 1/cosq • Drop at grazing incidence angle
Secondary Ion Mass Spectrometry (SIMS) Mass Ion beam detect sputtered species (neutrals, ions) from the sample S+ S • Sensitive to top most layers • Chemical composition • Structural informations • Very high sensitivity • Imaging: 100~1000nm • Depth profiling: 5nm • Ion yield depends on surface concentration and sputtering yield • Organic anlaysis: m/z = 5000~40,000 • Matrix effect: secondary ionization mechanism • Destructive: implantation, mixing, sputtering, ion beam induced • surface chemistry, radiation induced atomic redistribution
SIMS modes • Dynamic SIMS • high sputter rate • ~10 mA/cm2 • ~100 mm/hr • destructive • Depth profiling • Static SIMS • low sputter rate • ~1nA/cm2 • <10 Å/hr • nondestructive • Submonolayer analysis 1nA/cm2 =10-9A/cm2/1.6x10-19 C = 6.3x109 ions/sec-cm2 = 6.3x109 ions/sec-cm2 1015atoms/cm2 = 1.6x10-5 ML
Ionization methods: • electron impact • microwave • field ionization • laser ablation • Ion source • Ar+ ion • O2+: for electropositive elements • Cs+: for electronegative elements • Liquid metal: Ga+, In+ • - small beam size Instrumentation
Mass spectrometer • Quadrupole • inexpensive, compact • Double focusing electrostatic • /magnetic sector • high transmission • High mass resolution • Time of flight • -high molecular weight From Jeol
Imaging SIMS • scan ion beam or • ion detector • Beam size <10nm • Resolution ~100mm
Thermal desorption spectroscopyTemperature programmed desorpion -measure desorbing molecules by heating the surface using mass spectrometer Quadrupole mass spectrometer Adsorbed molecules heater • Heat of adsorption if Eads =Edes • Surface coverage: peak area • Adsorption sites: peak position • Intermolecular interaction • Kinetics of desorption : peak shape
Analysis of TPD Redhead, Vacuum 12, 203 (1963) The rate of desorption rd = -dq/dt = koqn exp(-Ed/kT) n: order of reaction ko : prexponential factor q : coverage Ed: activation barrier for desorption The sample temperature varies linearly T(t) = T0 + bt b = dT/dt : heating rate[K/s] rd = -dq/dT = (1/b)koqn exp(-Ed/kT) coverage kd=k0eEa/kT Intensity TPD spectra Temperature Ea = 24kcal/mol b= 10 K/sec n=1 ko=1013 1/sec Ed,ko’ q, b : Desorption temperature ko’ q,n: peak shape q: Peak area:
Zero-order desorption kinetics rd = -dq/dt = ko exp(-Ed/kT) • -independen of coverage • exponential increase with T • common leading edge • Rapid drop • Tmax move to higher T with coverage • Pseudo zerp-order for strong intermolecular • interactions between adsorbates Intensity T/K
First-order desorption kinetics n =1 rd = -dq/dt = koqexp(-Ed/kT) • -rate proportional to coverage • balance between q and exp(-Ed/kT) • Tpeak independent of q • Asymmetric line shape • Tpeak as Ed • Molecular desorption Intensity exp(-Ed/kT) q T/K
Second order desorption kinetics n=2 rd = -dq/dt = koq2 exp(-Ed/kT) • -rate proportional to coverage • balance between q2 and exp(-Ed/kT) • Tpeak varies with q • symmetric line shape • Common trail of peaks • Recomnative desorption • Pseudo-2nd order for strong • intermolecular interactions Intensity T/K
Fractional order desorption kinetics Indicate cluster formation on the surface Desorption from edge of clusters - indica Effect of activation barrier Ed=50~400kJ/mol Ed Intensity 10 20 30 40 50 Ed Tpeak peak width At saturation coverage Ed/RTp= 30kJ/mol