Aluminum
Explore the fascinating journey of aluminum from its ancient use to modern applications. Learn about its discovery by Thomas Dawson, improvements by pioneers like Oersted and Wohler, and its characteristic properties. Discover how aluminum is extracted from bauxite and its widespread utilization in aircraft, automobiles, and more. Delve into its atomic properties, classification as a metal, and its abundance in the Earth's crust. Uncover the significance of aluminum in various industries and daily life.

Aluminum
E N D
Presentation Transcript
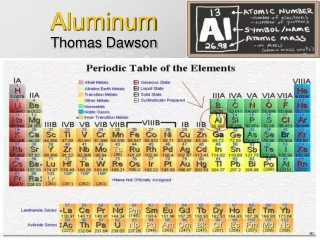
Aluminum Thomas Dawson
The Discovery • Aluminum was used by the Ancient Greeks and Romans, however they never identified it as Aluminum • Hans Christian Oersted (Denmark) • Developed first traces of Al from crystalline solid, alum (1825) • Rather impure though • Believed that Al existed in alum long before however no one had developed an extracting process • Reacted aluminum chloride with potassium metal in mercury • Friedrich Wohler (Germany) • Improved Oersted’s method and created a purer form (1827) • Henri Étienne Sainte-Claire Deville (France) • Further improved the extraction method which lead to its commercial production (1854) • Price:$1200/kg$40/kg
Characteristic Properties • How Scientists Know it’s Aluminum • Atomic Number:13 • Atomic Weight:26.9815386 • Melting Point:933.437 K (660.323°C or 1220.581°F) • Boiling Point:2792 K (2519°C or 4566°F) • Density:2.70 grams per cubic centimeter • Other Properties • Phase at Room Temperature: Solid Element • Reacts with acids to produce Hydrogen Gas • Classified as metal so… • Good Conductor • Ductile (Drawn into wire) • Lustrous (Bright silvery) • Malleable (Hammered into thin sheets)
Other Info • Period Number:3 • Group Number:13 • Group Name: none • From the Latin word for alum, alumen. • Aluminum is pronounced as ah-LOO-men-em • Classification: Metal • Sir Humphrey Davy proposed Aluminum as the name however he changed it to aluminium. In 1925 the ACS went back to the original spelling however the ret of the world still uses the –iumending
Where Does it Come From? • Is the most abundant metal in the earth’s crust (8%) • However it is so too reactive to be found isolated in nature • Found in a variety of minerals inc. clay and feldspar • Bound in tight compounds of Oxygen & Silicon • Main ore is mined in Jamaica, Australia, and Kansas-Bauxite • Aluminum Oxide-alumina-is extracted • Undergoes electrolysis process founded in 1886 by Charles M. Hill
How do we Use it Today • Because of its properties, Aluminum has a wide variety of ways in which it can be used • Many of which we use on a daily basis • Those Include… • Aircrafts • Electrical transmission wires (powerlines, cables, etc.) • Engines • Automobiles • Cooking utensils • Paint Pigment-makes the paint more waterproof and heat resistant • Aluminum Foil • Soda Cans • Lightweight containers • Softball/Baseball Bats
Bohr Rutherford, Electron Dot Diagram, and the Electron Configuration • Bohr Rutherford ModelLewis Dot Diagram Electron Configuration 1s22s22p63s22p1
Works Cited • Burns, Ralph A. "7.7 A Survey of Elements." Fundamentals of Chemistry. Fourth Edition ed. 2003. • Chemistry 448. Dept. home page. 29 May 2007. Marshall University. 29 May 2007 <http://science.marshall.edu/castella/chm448/elements1.pdf>. • Gagnon, Steve. "It's Elemental." Jefferson Lab. 28 May 2007 <http://education.jlab.org/itselemental/ele013.html>.