Biochemical Tests
Objectives. Tests to knowLactose fermentationIndole MotilityH2S production in SIMMethyl Red/Voges ProskauerCitrateOxidase test. Lactose Fermentation. MacConkey Agar contains bile salts and crystal violet, both inhibitory to Gram-positive bacteria and selects Gram-negative bacteria, such as


Biochemical Tests
E N D
Presentation Transcript
1. Biochemical Tests
2. Objectives Tests to know
Lactose fermentation
Indole
Motility
H2S production in SIM
Methyl Red/Voges Proskauer
Citrate
Oxidase test
3. Lactose Fermentation MacConkey Agar contains bile salts and crystal violet, both inhibitory to Gram-positive bacteria and selects Gram-negative bacteria, such as E. Coli. It also differentiates lactose-fermenting bacteria, such as E. Coli from non-lactose fermenting bacteria.
Media and Reagent: MacConkey Agar and neutral red dye
Method: Streak MAC plate and incubate at 37�C for 2 days.
Expected results:
Positive test: Lactose fermentation = Growth and color change to pink
Negative test: No lactose fermentation = May or may not grow and no color change
4. Enteric Gram-negative rods Lactose-fermenters (LFs)
Escherichia coli
Klebsiella pneumoniae
Non-lactose fermenters (NLFs)
Salmonella enterica
Shigella spp.
Proteus mirabilis
Pseudomonas aeruginosa
5. Results of Lactose Fermentation
6. Indole Production Test The amino acid tryptophan can be broken down by enzyme tryptophanase to form indole, pyruvic acid and ammonia as end products. Tryptophanase differentiates indole-positive enterics, such as E. coli and P.vulgaris from indole-negative enterics, such as S. marcescens.
Media and Reagents: SIM with tryptophan and Kovac�s reagent.
Method: Inoculate medium and incubate at 37�C for 24-48 hours. After incubation, add five drops of Kovac�s reagent to the surface. Do not stir or shake the tube.
Expected Results:
Positive test : Kovac�s reagent combines with indole and turns the surface red.
Negative test: No red color development
7. Motility Test This is not a biochemical test, but it can distinguish bacteria. It determines presence of flagella.
Media and reagent: Deep agar
Method: Inoculate deep with a needle and incubate at 37�C for 24-48 hours.
Expected results:
Positive test: Growth spread away from the line of inoculation = motile
Negative test: Growth only occurred at the line of inoculation = Non-motile
8. Results for Motility Test
9. Indole Production
Purpose: The indole test is used to identify bacteria capable of producing indole using the enzyme tryptophanase.
Kovac�s Reagent
10. Results of H2S production
11. Sulfur Reduction
Purpose: This test is used to identify those bacteria capable of reducing sulfur.
12. . SIM Tube Results A. Indole positive and hydrogen sulfide positive
B. Hydrogen sulfide positive
C. Indole positive and motility positive (note fuzzy growth away from stab line)
D. Negative Control
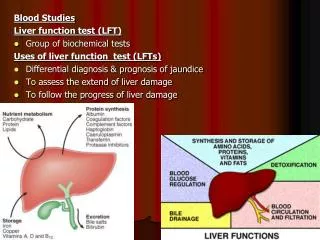
13. Methyl Red Test Methyl red test is used to identify enteric bacteria based on their pattern of glucose metabolism. If they use mixed acid pathway and produce acidic products, then they are called methyl-red-positive. If they use butylene glycol pathway and produce neutral end products, then they are called methyl-red-negative.
Media and reagents: MR-VP medium and methyl red indicator
Method: Inoculate broth and incubate at 37�C for 2-5 days. After incubation, transfer 2.5 ml of inoculate to another tube and add five drops of methyl red. Roll between the palms of hands to disperse methyl red.
Expected results:
Positive test: acids + methyl red = red solution
Negative test: neutral end products + methyl red = yellow color
14. Results for Methyl Red Test
15. Methyl Red (MR) Test Purpose: Used to identify bacteria that produce stable acid end products by means of mixed acid fermentation of glucose
16. Voges Proskauer Test It is used to identify enteric bacteria based on their pattern of glucose metabolism. The enterics that produce neutral end-products, such as acetoin are detected by VP test.
Media and Reagent: MR-VP medium and Barritt�s Reagent A (contains alpha-naphthol) and Barritt�s Reagent B (contains KOH).
Method: Inoculate medium and incubate at 37�C for 48 hours. After incubation, transfer 2.5 ml of inoculate to another tube and add six drops of Barritt�s Reagent A and two drops of Barritt�s Reagent B. Gently mix and let it sit for 10-15 minutes to allow time for color development.
Expected results:
Positive test: acetoin + alpha-naphthol + KOH = red color
Negative test: alpha-naphthol +KOH = copper color
17. Results for VP test
18. Citrate Utilization Citrate is an organic molecule that can be utilized by bacteria that produce the enzyme citrase. Citrase is produced by some bacteria such as E. aerogenes but not by others like E. Coli
Media and Reagent: Simmon�s Citrate Agar. It has citrase as the only carbon source and PH indicator bromothymol blue
Method: Inoculate the slant and incubate at 37�C for 24-48 hours.
Expected results:
Positive test: Growth and color changes to blue
Negative test: No growth and color remains green
19. SIMMONS CITRATE Purpose: The citrate utilization test is used to determine the ability of an organism, using the enzyme citrase, to use citrate as its sole carbon source
21. IMViC Reactions I M Vi C
Escherichia coli + + � �
Proteus vulgaris + + � �
Klebsiella pneumoniae � � + +
Enterobacter spp. � � + +
Serratia marcescens � � + +
Citrobacter freundii � + � +
Citrobacter koseri + + � +