CARBON DIOXIDE
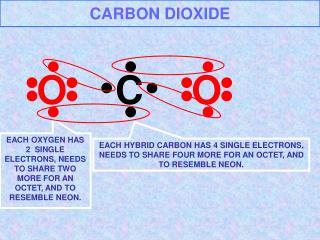
CARBON DIOXIDE. C. O. O. EACH OXYGEN HAS 2 SINGLE ELECTRONS, NEEDS TO SHARE TWO MORE FOR AN OCTET, AND TO RESEMBLE NEON. EACH HYBRID CARBON HAS 4 SINGLE ELECTRONS, NEEDS TO SHARE FOUR MORE FOR AN OCTET, AND TO RESEMBLE NEON. CARBON DIOXIDE, VENN DIAGRAM. O. C. O.

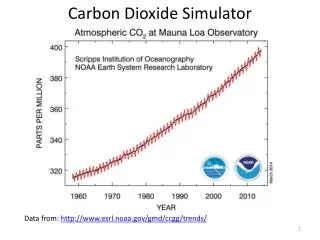
CARBON DIOXIDE
E N D
Presentation Transcript
CARBON DIOXIDE C O O EACH OXYGEN HAS 2 SINGLE ELECTRONS, NEEDS TO SHARE TWO MORE FOR AN OCTET, AND TO RESEMBLE NEON. EACH HYBRID CARBON HAS 4 SINGLE ELECTRONS, NEEDS TO SHARE FOUR MORE FOR AN OCTET, AND TO RESEMBLE NEON.
CARBON DIOXIDE, VENN DIAGRAM O C O THE YELLOW CIRCLE REPRESENTS THE OXYGEN”SET” WITH AN OCTET THE GREEN INTERSECTION AREAS REPRESENT THE 4 SHARED ELECTRONS OF A DOUBLE BOND THE BLUE CIRCLE REPRESENTS THE CARBON”SET” WITH AN OCTET
CARBON DIOXIDE, VENN DIAGRAM O C O CO2 HAS AN IONIC CHARACTER OF 0.9, WHICH IS CONALENT POLAR AS WIH ALL COVALENT COMPOUNDS, CO2 HAS WEAK ATTRACTIONS, LOW M.P. AND B.P.AND IS NEVER CONDUCTIVE. EACH DOUBLE BOND IS 4 SHARED ELECTRONS, IS SHORTER AND STRONGER THAN THE SINGLE BOND.