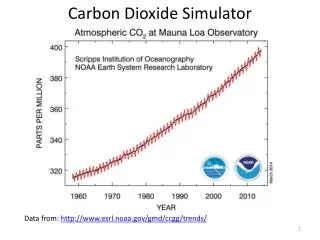
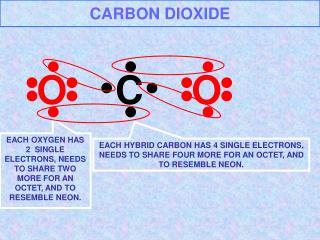
Carbon Dioxide
Carbon Dioxide. Icy Chan F.3D (2). History of carbon dioxide. Scottish physician Joseph Black. The Flemish chemist Jan Baptist van Helmont. Carbon dioxide (chemical formula CO 2 ) is a chemical compound composed of two oxygen atoms covalently bonded to a single carbon atom. .

Carbon Dioxide
E N D
Presentation Transcript
Carbon Dioxide Icy Chan F.3D (2)
History of carbon dioxide. Scottish physician Joseph Black. The Flemish chemist Jan Baptist van Helmont
Carbon dioxide (chemical formula CO2) is a chemical compound composed of two oxygen atoms covalently bonded to a single carbon atom.
Molar mass: 44.0095(14) g/mol • Molecular shape: linear • Test the presence of carbon dioxide: turn limewater from colourless to milky • Other names: Carbonic acid gas; carbonic anhydride; dry ice
Carbon dioxide pressure-temperature phase diagram showing the triple point and critical point of carbon dioxide.
laboratory preperation: • In the laboratory, carbon dioxide is usually prepared by the action of dilute hydrochloric acid on marble chips. Marble chips are taken in a round-bottomed flask, as shown in figure 11.6. • Dilute hydrochloric acid is added through the thistle funnel. The gas can be collected by the upward displacement of air.
Uses of carbon dioxide Food Beverages Wine making Fire extinguisher Lasers Refrigerant pH control Oil recovery