Solid Carbon Dioxide
Solid Carbon Dioxide. By: Hayley and Mae. Molecular solids. Solids made of separate molecules packed together are known as discrete molecular substances . From the word molecular means composed of molecules. There are two forces present in discrete molecular substances.

Solid Carbon Dioxide
E N D
Presentation Transcript
Solid Carbon Dioxide By: Hayley and Mae
Molecular solids • Solids made of separate molecules packed together are known as discrete molecular substances. • From the word molecular means composed of molecules. • There are two forces present in discrete molecular substances. • The forces within the molecule-intramolecularforces-are the covalent bond linking atoms in individual molecules. • Covalent bond consist of shared paired of electrons. These electrons must exist between the atoms sharing them. • The forces between the molecules-intermolecularforces-link individual molecules to each other. • Intramolecular forces are very strong. • Intermolecular forces are much weaker than intramolecular forces. • Intermolecular forces determine the physical properties of discrete molecular substances. • Polar molecular substances are generally soluble in water. • Non-polar molecular substances are generally insoluble in water.
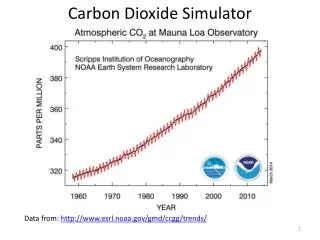
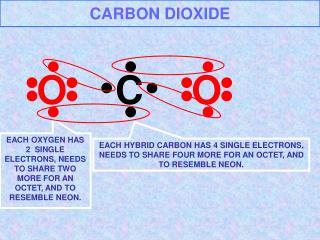
Solid Carbon Dioxide Solid Carbon Dioxide (dry ice) is a solid at room temperature and sublimes (turns directly into gas) because the forces between the molecules (ie.The intermolecular forces) are very weak. The carbon dioxide molecule (O=C=O) contains two double bonds, 2 hydrogen atom bonded to a single carbon atom and has a linear shape.
When the substance sublimes the intramolecular forces (ie. Covalent bond) remain intact. The molecules do not break down- they simply separate between molecules due to heat energy. • The regular arrangement of molecules are held together by weak forces. • Solid Carbon Dioxide is made entirely of molecules. • The atoms are non-metal atoms that is covalently bonded together. • Separate molecules are linked together by weak van der Waals forces (VdW). • VdW’s forces result from long range attraction of electrons in 1 molecule for protons in an adjacent molecules & vice versa. • The low temperature and direct sublimation to a gas makes dry ice an effective coolant (a fluid that flows through a device to prevent from over heating), since it is colder than water ice and leaves no residue as it changes state. • Solid carbon dioxide or also known as dry ice is non-polar,which means it is symmetrical and cancel each other out, and with a dipole moment of zero, so attractive intermolecular van deer Waals forces operate. The composition results in low melting and boiling point temperature and electrical conductivity.
Properties of Solid Carbon Dioxide • Melting and Boiling point • Low melting point (mp) & Boiling point (bp) • Soft • Electrical Conductivity • Electricity is flow charged particles. • Since Solid Carbon Dioxide is consists of molecules and not charged particles they do not conduct electricity (no free electrons of ions) • Solubility of Solid Carbon Dioxide • Carbon Dioxide is non-polar molecular solids so this substance only dissolves in non-polar solvents, because as you know in the molecular solid solubility property “non-polar molecular substances only dissolved in non-polar solvents and insoluble in water”.