The Periodic Table
240 likes | 455 Vues
The Periodic Table. Chemistry Chapter 6. Organizing the Elements. Chemists first used the properties of elements to sort them into groups. First classification system (1829) involved grouping know elements into triads . Didn’t work for all known elements.

The Periodic Table
E N D
Presentation Transcript
The Periodic Table Chemistry Chapter 6
Organizing the Elements • Chemists first used theproperties of elements to sort them into groups. • First classification system (1829) involved grouping know elements intotriads. Didn’t work for all known elements. • In the mid-1800s another system was proposed, by a Russian Chemist Dmitri Mendeleev.
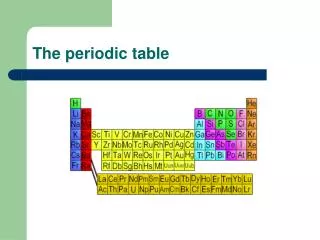
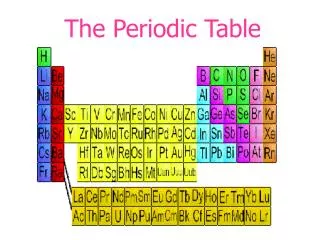
Mendeleev’s Periodic table • He arranged elements in a periodic table. • Elements in a periodic table are arranged into groups based on a set of repeating properties. Mendeleev arranged the elements in his table in order of increasing atomic mass. • He left spaces in his table for yet undiscovered elements and was even able to predict their properties.
Problem with Mendeleev’s Table • What was wrong with Mendeleev’s table? • 50 years after Mendeleev, another scientist named Henry Mosley corrected the order of the elements. • Mosley determined the atomic numbers of all the elements and arranged them based on their atomic numbers. • This allowed elements with similar physical and chemical characteristics to fall into place without exception.
Periodic Law(Mendleev’s Law) When elements are arranged in order of increasing atomic number, there is a periodic repetition of their physical and chemical properties. • Elements in a group have similar properties!!!!!!!Why?? • Same # of Valence Electrons!
Metals, Nonmetals, & Metalloids • Elements can be classified into three broad classes based on their general properties. • Metals – good conductors, high luster, ductile, and malleable. Most elements are metals. • Nonmetals – great variation in physical properties. In general have properties opposite of the metals. • Metalloids – has properties of both metals & nonmetals.
Element Key Atomic number 2 4 Electrons in each Energy level 6 C Element symbol Element name carbon 12.011 Atomic mass
Electron Configurations & Groups • Elements can be sorted into noble gases, representative elements, transition metals and inner transition metalsbased on their electron configurations. • With the exception of the transition metals (Group B), the group # lets you know how many electrons are in the outer energy level (# of valence electrons).
The Nobel Gases • The noble gases (inert gases) are the elements in Group 8A of the periodic table. • S & P sublevels are completely filled with electrons. • Due to filled outer energy level, noblegases do not readily react with other elements. • Octet Rule – atoms are most stable when they have 8 electrons in their out level.
The Representative Elements • Elements in groups 1A through 7A are often referred to as representative elements because they display a wide range of physical & chemical properties. • In atoms of rep. elements, the s & p sublevels of the highest occupied energy level are not filled. • Its group number is equals the number of electrons in the highest occupied energy level.
Transition Elements • Elements in the B groups, which provide a connection between the two sets of rep. elements, are referred to as transition elements; two types of transition elements. • Transition metals – the highest occupied s sublevel & nearby d sublevel contain electrons. These elements are characterized by electrons in the d orbitals. • Inner transition metals(rare earth elements) – The highest occupied s sublevel and nearby f sublevel generally contain electrons. Characterized by f orbitals that contain electrons.
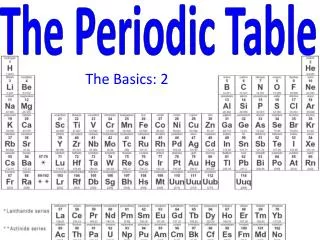
Periodic Trends • Trends in Atomic Size • Atomic radius is one half of the distance between the nuclei of two atoms of the same element when the atoms are joined. In general, atomic size increases from top to bottom within a group and decreases from left to right across a period.
Atomic size increases in groups as atomic number increases. • Atomic size decreases across a period from left to right.
Ions • Some compounds are composed of particles called ions. • An ion is an atom or group of atoms that has a + or – charge. How does this occur? • + or – ions form when electrons are transferred between atoms. • An ion with a + charge is called acation. • always smallerthan the atoms from which they form • An ion with a – charge is called ananion. • always larger than the atoms from which they form.
Trends in Ionization Energy • If an atom is subject to enough energy electrons may be removed; this is referred to as ionization energy. • The energy required to remove the 1st electron from an atom is called 1st ionization energy. First ionization energy tends todecrease from top to bottom with in a group and increase from left to right across a period.
Trends in Electronegativity • Electronegativityis the ability of an atom of an element to attract electrons when the atom is in a compound (electron affinity). • Used to predict the type of bond that will form during a chemical reaction. In general, the electronegativity values decrease from top to bottom within a group. For representative elements, the values tend to increase from left to right across a period.
Summary of Trends Atomic Radius trend: Largest toward SW corner of PTIonization Energy trend: Largest toward NE of PTElectronegativity : Most favorable NE of PT