Enzymes
Enzymes. Proteins acting as enzymes!. What is an enzyme?. Most enzymes are either tertiary or quaternary proteins Enzymes act as a catalyst in a chemical reaction Speed up the reaction without being permanently changed in structure. More about enzymes….

Enzymes
E N D
Presentation Transcript
Enzymes Proteins acting as enzymes!
What is an enzyme? • Most enzymes are either tertiary or quaternary proteins • Enzymes act as a catalyst in a chemical reaction • Speed up the reaction without being permanently changed in structure
More about enzymes… • Because they are not permanently changed, they are reusable • Enzymes are specific to what reactions can be catalyzed
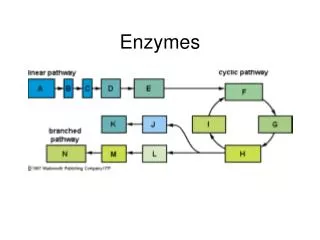
Names of enzymes • End with –ase and are named based on what they catalyze • Sucrase helps to hydrolyze sucrose • Oxidase helps to catalyze an oxidation reaction • Reductase helps to catalyze a reduction reaction • Dehydrogenase removes hydrogen atoms • Protease hydrolyzes peptide bonds • Nuclease hydrolyses nucleic acids • Synthase combines two molecules
How do they work? • Enzymes lower the activation energy of a reaction • This allows the reaction to take place at a faster rate and with much less energy being used • Example: hydrolysis of proteins in our diet would happen without a catalyst, but not fast enough to meet the body’s requirement for the amino acids
Without Enzyme With Enzyme Free Energy Free energy of activation Reactants Products Progress of the reaction Enzymes
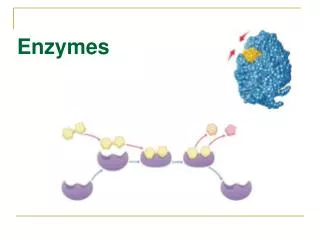
Enzyme Enzyme “parts” Active Site • What makes enzymes selective is their unique 3D shape that recognizes and binds a small group of reacting molecules, called SUBSTRATES • Where the enzyme binds that substrate is called the ACTIVE SITE Substrate
Two types of “fits” • Lock-and-key model • Active site is rigid and nonflexible • Induced Fit • There is an interaction between the enzyme and substrate to give the enzyme a broader range of activities
Environmental Conditions/Factors that affect enzyme activity • Temperature • Too low – not enough energy for catalysis • Too high temperatures – cause denaturation of proteins • pH • Also should be at the optimum pH (most are in the range of 6-8) • Substrate Concentration
Enzyme Inhibition • Inhibitors cause enzymes to lose catalytic activity • Competitive inhibition • Noncompetitive inhibition
Enzyme Competitive Inhibitors • Has a structure similar to the substrate • Competes for the active site, making it so the substrate cannot bind Substrate
Noncompetitive Inhibitors • Does not bind to the active site, rather elsewhere on an enzyme • This changes the shape of the enzyme, making it so the substrate cannot fit properly
Enzyme Cofactors • When enzymes require the presence of another molecule or ion in order to catalyze properly • Metal ions • Example: hemoglobin needs iron present in order to bind to oxygen molecules • Vitamins