Single ventricle
Single ventricle. Motasem is a 7 years old male patient born at home in Palestine as a product of vaginal delivery with mild cyanosis of the lips and extremities. At the age of 3 months the cyanosis became more prominent, so the family seeks medical advice.

Single ventricle
E N D
Presentation Transcript
Motasem is a 7 years old male patient born at home in Palestine as a product of vaginal delivery with mild cyanosis of the lips and extremities. • At the age of 3 months the cyanosis became more prominent, so the family seeks medical advice. • The patient was diagnosed to have congenital heart disease (single ventricle) and the family informed that there is no good facility to treat such a case.
After that the patent developed recurrent chest infection shortness of breath, poor gain weight, poor feeding . • At the age of 2 years patient underwent surgical intervention by Belgian doctor (glenn shunt) and. the general condition of the patient was improved partially.
PMH • Recurrent admission with chest infection. • Feeding :breast feeding 1 year, added food:6 months . • Development /poor gain weight, walk:1 year, talk :1 year • Good performance in first year school. • Family history :no family history of similar condition, have 4 sibling, all are well. • Drug history /not on chronic medication. no known drug allergy.
ON EXAMINATION • The patient lying flat, conscious alert active, oriented for time ,place,person,not pale ,not jaundiced or cyanosed • V/S: pulse :100per min ,R.R/ 20 Tempreture:37.4 AX BP:100/60 • Weight:14kg(2 stander deviation below 5%),length:108cm(3 stander deviation below 5%) • Head &neck /free • Chest/good air entry bilateral, no added sounds. • Heart/ejection pan systolic murmur, systolic thrill • Abdomen/soft lax, no mass • CNS Grossly intact.
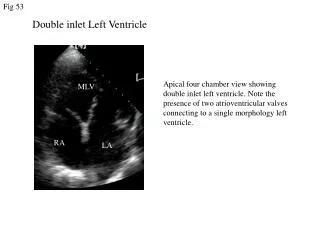
Investigation • cardiac cath:douple inlet left ventricle with transposition of great vessels ,sever sub pulmonary stenosis. • Hb:15.7 mg/dl,WBC:5.9 NEUTRO:60% ,PLT:238, PT:13.3 ,PTT:32.4, • INR:1.03 UREA:22, CREAT:0.22, LFT:NORMAL(ALB:4) ,KFT:NORMAL
1) p wave are normal ,spiked,or bifid. • 2)precordial lead pattern suggests right ventricular hypertrophy,combined ventricular hypertrophy,or sometimes left ventricular dominance.
On 14/12/2003 patient underwent Gortex graft after that patient was transferred to the adult ICU for 5 days then he transferred back to the floor with bilateral chest tube and he was on: • Warferin,albumin,asprin,cagluconate,lasix, ceftriaxon,IVF 2/3 maintenance. • The patient stay in our floor from 19/12/2003to 24/2/2004.
During these period patient developed electrolyte disturbance ,hypoalbuminemia,coagulopathy and received blood,FFPseveral times • on 22/12 blood culture done revealed gram – rods(acetenobacter boumani) sensitive to fortum (1 week) • swab culture done revealed staph epidermidies sensitive to vancomycine (1 week). • On 3/1/2004 cardiac cath done revealed grade 1-2 tricuspid regergitation,mild diastolic disfunction. So enalopril was added.
On 19/1/04the left chest tube was removed.after that the patient developed minimal plural effusion on the left side. • On 10/2/2004 patient developed hepatomegaly a bout 3 cm BCM,abd u/s was done revealed enlarged liver without evidance of focal lesions,minimal ascites. • On 17/2/2004 the right chest tube was removed. • Patient was discharged in a good condition on 24/2/2004 on: • Sopak,warferin,lasix,asprin,enalopril,lanoxine, • moduritic. • Plan: follow up with cardiologist in palastine
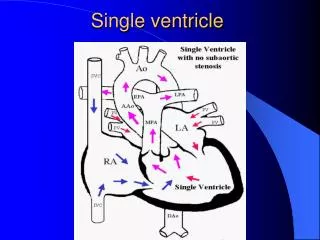
Single ventricle • both atria connect to the single ventricle via either a common atrioventricular valve (sometimes termed the common-inlet ventricle) or separate atrioventricular valves (double-inlet ventricle) with total mixing of systemic and pulmonary venous returns. Single ventricle most frequently coexists with transposition. Subpulmonary stenosis is more prevalent than the combination of aortic arch obstruction and subaortic stenosis. single ventricle can occur without stenosis of either pulmonary or aortic outflow
The embryology of single ventricle in humans is still unknown. Presumably, both ventricular septation and movement of the common atrioventricular orifice are disrupted. It is likely that many genetic alterations can result in a single ventricle phenotype.
Pathophysiology: In fetal development, there is no circulatory derangement since pulmonary circulation and systemic circulation are normally in parallel with 2 levels of connection: atrial and ductal. Lack of separation between pulmonary and systemic circulations, however, causes obvious cyanosis postnatally, with severity dependent on the degree of coexistent subpulmonary stenosis. Although cases of single ventricle and arch obstruction are the least cyanotic since they never display subpulmonary stenosis, such patients are vulnerable to poor lower body perfusion upon reduction in ductal diameter
Frequency: • In the US: Single ventricle occurs in approximately 5 of every 100,000 live births • Mortality/Morbidity: The severity and timing of presentation depend on the extent of coexistent subpulmonary stenosis (or, alternatively, aortic obstruction) and on reduction in caliber of the ductus arteriosus. • Sex: No known disparities exist. • Age: Presentation is generally within the first month of life. As the ductus arteriosus reduces in caliber within the first few days of life, those infants with severe subpulmonary stenosis or aortic obstruction present with cyanosis or poor peripheral perfusion, respectively
CLINICAL manifestation • The clinical picture is variable depending on the associated intracardiac anomaly. • If pulmonary out flow is obstructed ,the presentation may be similar to that of tetralogy of fallot.(marked cyanosis without heart failure) • If pulmonary out flow is not obstructed the presentation is similar to that of transpostion with VSD:minimal cyanosis with marked heart failure. • Neonates with single ventricle and aortic obstruction may have rapid breathing, lethargy, and poor feeding.
Physical: • The first heart sound is normal. • The second heart sound is single and load.. • A 3 ed heart sound is common and may be followed by a short mid-diastolic rumbling murmur caused by increased flow through the atrioventricular valves. • ejection systolic murmur is present in those with subpulmonary stenosis as well as those with aortic obstruction. • Systolic thrill. • Left parasternal lift is palpable
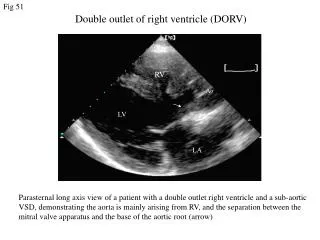
DIFFERENTIALS • Aortic Stenosis.Corrected Transposition of the Great Arteries: Double Outlet Right Ventricle:Hypoplastic Left Heart Syndrome • Neonatal Sepsis [Protein-losing Enteropathy]Pulmonary Stenosis
Glenn shunt:is an anastamosis between the superior vena cava and the pulmonary arteries (RPA). • The benefit of glenn shunt is that it reduces the work on the left ventricle and may lessen the chances acquiring left ventricular dysfunction later in life. • Modified fontan operation:this procedure was performed by anastomosing the right atrium or atrial appendage directly to the pulmonary artery
Currently, a modification, known as a cavopulmonary isolation procedure,is performed. • This involves anastomosing the inferior vena cava to the pulmonary arteries either via a baffle that runs along the lateral wall of the right atrium or via a homo graft or Gore Tex tube running outside the heart.. • The advantage of this approach is that blood flows by a more direct route into the pulmonary arteries, decreasing the possibility of right atrial dilatation and markedly reducing the incidence of post operative plural effusions which were common with the earlier method.