The Single Ventricle
The Single Ventricle. Karim Rafaat, M.D. The title “ single ventricle ” includes those lesions designated as both HLHS HRHS HLHS is far more common, and the strategy for palliation of both lesions similar, so I will not mention HRHS. HLHS - History.


The Single Ventricle
E N D
Presentation Transcript
The Single Ventricle Karim Rafaat, M.D.
The title “single ventricle” includes those lesions designated as both • HLHS • HRHS • HLHS is far more common, and the strategy for palliation of both lesions similar, so I will not mention HRHS
HLHS - History • First described in 1952 by Lev as the pathologic complex “hypoplasia of the aortic tract”,included cases of: • hypoplasia of the aorta and VSD • hypoplasia of the aorta with aortic stenosis or atresia, with or without mitral stenosis or atresia • In 1958, Noonan and Nadas termed these lesions as “hypoplastic left heart syndrome”.
Embryology • The embryologic cause is not fully understood. • It probably results from a limitation of either LV inflow or outflow, such as the development of severe AS early • Decreased antegrade flow through LV • most common cause is mitral atresia • decreased division of cardiac myocytes
Genetics • Familial inheritance: • Autosomal recessive and multifactorial inheritance have both been postulated. • Sibling recurrence risk: 0.5% • Sibling recurrence for all other cardiac malformations: 2.2% • Definable genetic disorder (28%): • Turner Syndrome • Noonan Syndrome • Trisomy 13, 18, 21, or other microdeletion syndromes
Epidemiology • Uniformly lethal prior to 1980 • Each year, approximately 1000 infants with HLHS are born in the US. • Prevalence: 1 per 6000-7000 live births. • In pathologic series, it accounts for 1.4-3.8% of congenital heart disease. • Third most common cause of critical CHD in the newborn. • 23% of all neonatal mortality from CHD • Male predominance: 57-70%.
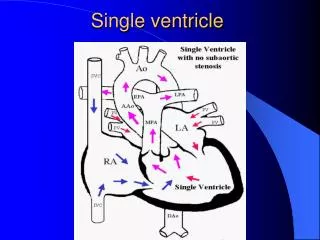
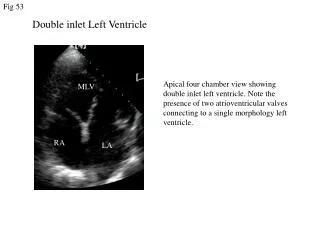
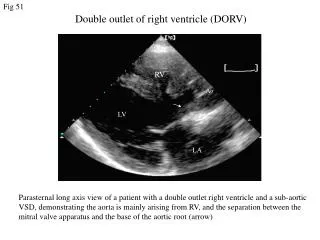
Anatomy • Underdevelopment of the left side of the heart • atresia of the aortic or mitral orifice • hypoplasia of the ascending aorta. • The left ventricle may be small and nonfunctional or totally atretic
Pulmonary venous return from LA to RA through a large PFO or ASD • Systemic venous blood mixes with pulmonary venous blood in the RA and RV • RV ejects blood into a large MPA
Systemic circulation is supplied in parallel with pulmonary circulation through a PDA • Multiple obstructions to systemic flow • Aortic valve atresia • Arch hypoplasia • Place systemic flow at risk
Blood flow to the coronary and cerebral circulations is retrograde • Usually little or no flow through aortic valve • Postnatal decline in PVR • places systemic, and especially the ductal dependant and retrogradely supplied coronary and cerebral vascular beds at risk for hypoperfusion secondary to pulmonary run-off
Pathophysiology • Relative Qp and Qs determined via resistances of respective vascular beds • Ventricle must supply both Qp and Qs • Single right ventricle has at least twice the volume load of an in series ventricle • Significantly volume overloaded
The aim of initial management is to optimize Qp and Qs in a manner that provides adequate end organ oxygen delivery without overloading the single ventricle • Remember my last lecture? • This balancing act is only temporizing and serves to allow pt to survive to definitive treatment
Treatment options • Supportive care • Only option up to 25 years ago • Is still main option of treatment in many countries • Staged reconstruction • Stage I Norwood Procedure • Stage II Bi-directional Glenn or Hemi-Fontan • Stage III Fontan Procedure • Transplant
Goals of Surgery • Unobstructed systemic blood flow • To maximize oxygen delivery and minimize ventricular hypertrophy • Limited pulmonary blood flow • To minimize ventricular volume load and the risk of pulmonary hypertension • Unobstructed pulmonary venous return • To minimize secondary pulmonary artery hypertension • Minimize likelihood of pulmonary artery distortion • Avoid dysrhythmias
All these goals, achieved in a timely fashion, circumvent the major risk factors for poor outcome post-Fontan: • Ventricular hypertrophy causing diastolic dysfunction • Elevated PVR or pulmonary artery pressure • AV valve regurgitation • Ventricular systolic dysfunction • The reasons why the above hurt the post-fontan heart will be discussed later
Stage I – Norwood palliation • The goal of the Norwood is to stabilize and balance the parallel circuit, protect the pulmonary vascular bed and preserve ventricular function • Adequate oxygen delivery allows for the growth necessary for a hemi-fontan or BDG to be performed
Native ascending and transverse aortic arch is incorporated into a neo-aorta • Neo-aorta created by augmenting native arch with autologous pulmonary homograft • Neo-aorta is attached to the proximal pulmonary artery trunk • Neo-aorta provides systemic outflow • Important that the neo-aorta is free of obstruction • Obstruction is poorly tolerated by the single ventricle and is associated with increased interstage mortality
Distal MPA is closed • Pulmonary flow is provided by a restrictive shunt from the right innominate artery to the RPA • Modified BTS
Post-Norwood issues • The hope is that now • RBTS + Rp = Rs • So the circulations are balanced and volume work is minimized • Meaning for a given required Qs, total Q can be less as the ratio is more favorable • But……
The ventricle has just been through hypothermic cardiopulmonary bypass with myocardial ischemia/arrest • Vascular endothelium of the systemic and pulmonary circulations have also been subjected to bypass and injury • Combined effect is a systemic inflammatory and adrenergic stress response • The ventricle can also exhibit a low cardiac output syndrome in the first 12-24 hours post op
All vascular beds show signs of endothelial dysfunction • Evidenced by increased resistance • This may tip the balance of flow towards the pulmonary circulation • Systemic oxygen demands may be unable to be met by the post-op ventricle • Leading to anaerobic metabolism, acidosis and worsening function • LCOS
Low Cardiac Output • Low systemic cardiac output can be due to • Globally decreased ventricular function • Elevated Qp:Qs • AV valve regurgitation • How to discern between the above?
Echocardiography • Evaluates pump function and rules out AV valve regurg • Arterial-venous oxygen saturation difference • An A-V DO2 more than 40% suggests inadequate tissue delivery of oxygen and low systemic cardiac output • OR…Lactate level plus base deficit • Good echo function plus high A-V DO2 = Qp>Qs
Treatment • One must pay attention to both TOTAL CO and the Qp:Qs ratio • The ratio can be altered by maneuvers discussed in my last talk • Total CO can be increased by careful selection of vasoactive agents • Want to avoid tachycardia and increasing afterload • Milrinone • Nesiritide • Dopamine
Hypoxemia • Pulmonary Venous desaturation • Atelectasis • pulmonary edema • pneumothorax • Systemic venous desaturation • Anemia • Low cardiac output • Decreased pulmonary blood flow • Elevated PVR • Pulmonary venous hypertension • Pulmonary artery distortion • Restrictive systemic to pulmonary shunt • Gotta rule out the top two, then, think about echo or cath to rule out the anatomic causes • Which need a surgeon….
Coronary circulation • Single right ventricle coronary blood flow occurs predominantly in diastole • Like an in series LV • When pulmonary flow is supplied by a shunt from a systemic artery, increases in SVR lead to increased pulmonary flow, and increased diastolic pulmonary run-off • This can lead to myocardial ischemia….and sudden death • Which is why
Dr. Cocalis The Wall of Wo “Leaving a kid in Norwood physiology is like taking a walk through Watts at midnight”
Sudden death post Norwood • Unpredictable and sudden • Experienced centers report survival between 63-94%1 • Inter-stage mortality of 10-15%2 • Rapid fall in PVR, or increase in SVR • Steal from coronary arteries • lower pressure in pulmonary circulation throughout cardiac cycle The Journal of Thoracic and Cardiovascular Surgery 2003;126(2) 504-509 Arch Dis Child Fetal Neonatal Ed 2005;90:F97-102.
Bartram et al, Causes of Death after the Modified Norwood procedure: A study of 122 postmortem cases, Ann Thorac Surg, 1997 • 122 cases over 15 years • The leading causes of death • largely correctable surgical technical problems associated with perfusion of the lungs (36%), of the myocardium (27%), and of the systemic organs (14%).
The proposed solution to • surgical manipulation of the coronary arteries • the pulmonary diastolic run-off through the modified BTS • Is an RV to PA conduit • First described by Norwood in 1981 • Reintroduced by Japanese surgeon Sano in the late 1990’s
The Sano modification • Directly supplies pulmonary flow via the RV • Aortic diastolic runoff does not occur • Post-op diastolic BP is higher • Coronary perfusion is improved • Blood flows only during systole • Reducing total pulmonary blood flow • Improves Qp:Qs, thus protecting pulmonary vascular bed and decreases volume load on the RV, giving it a greater chance to return to normal size and function
Less distortion of the pulmonary arteries than is seen with a BTS • Improved growth of PA’s
Trade off’s • Ventriculotomy • Increases potential for low cardiac output syndrome • The damage to the ventricular wall may be offset by the better coronary perfusion….. • Increased volume load secondary to reversed diastolic flow in a non-valved conduit • Possibility of shunt occlusion • Concern of RV arrhythmias post ventriculotomy • Not confirmed by present studies, though
Januszewska et al, RV to PA shunt and modified BTS in preparation for hemi-Fontan procedure in children with HLHS, European Jour Cardiac Surg, 27, 2005 • 78 children – 27 underwent Norwood with BTS 51 underwent Sano modification • Those who underwent Sano, at time of hemi-fontan • Larger pulmonary arteries • Which means lower resistance to the passive flow that will be supplying the lungs after the BDG or Fontan • Less RVH • Better diastolic function, and so lower filling pressures required • Lower Qp:Qs (0.8 vs 1.27) • Less pulmonary vascular remodeling and less ventricular volume load
Pizzaro et al, Right Ventricle to Pulmonary Artery Conduit Improves Outcome after stage I Norwood for HLHS, Circulation, 2003; 108 • Retrospective cohort review • 36 RV to PA conduits • 20 BTS • Those with RV to PA conduits • Higher diastolic BP • Lower PaO2 • Indicating lower Qp:Qs secondary to less diastolic run-off • Less ventilatory manipulations were required for Qp:Qs management • 33/36 survived to BDG vs 14/20 in the BTS group
Other Considerations • Risk for shunt occlusion • Low sats lead to high Hct’s, which increases risk of thromboembolic complications • Need to be well hydrated • Shunt failure • Slowly occurs as pt grows, but shunt does not • Leads to slowly progessive cyanosis as oxygen consumption increases in a growing pt • VENOUS ACCESS • Any venous embolus may reach systemic vascular beds • Watch for air bubbles, clots, meticulously….
Glenn Hughes – A Village Person Bidirectional?
Stage II – Partial Cavopulmonary Anastomosis • After stage I, there are two problems • Cyanosis • Excessive ventricular volume load • The Fontan fixes both of the above, but must come after an intermediate step… • Why?
Reasons for a staged repair • The fontan requires low PVR to allow for passive pulmonary flow • PVR does not reach nadir until 6-8 months • Furthermore, following the high Qp:Qs state of pre-norwood, the pulmonary vasculature can be reactive • Which is exacerbated by the stress of bypass
The parallel circulation single ventricle is relatively hypertrophied and dilated secondary to volume overload • Shifts Frank-Starling curve down and to the right • Means the norwood ventricle is very volume sensitive • A loss of ventricular filling secondary to increases in PVR would lead to critically decreased CO
The solution is a staged procedure that allows for more gradual ventricular unloading and remodeling • Also allows for adjustment of the upper body venous and lymphatic systems to deal with an increase in venous pressure prior to the Fontan • Usually performed around 4-6 months of age
Bidirectional Glenn • The RV/PA or BT shunt is removed • This volume unloads the ventricle • Critical in improving outcome in single ventricle palliation • SVC is anastomosed end to side with the RPA • Is more compatible with an extracardiac fontan procedure down the line
Hemi-Fontan • Similar to BDG physiologically • Has additional proximal SVC and inferior RPA anastomosis • RA communication closed with a patch • More suited for eventual lateral baffle Fontan
Stage II Physiology • Half the blood to the heart comes from the IVC, half from the pulmonary veins • Qp:Qs is now 0.5 • SaO2 about 75-85% • Infants with bigger heads have higher sats • Excessive volume load is now eliminated • Ventricle now pumps only Qs • Decreased cavity dimension and increased wall thickness improves tricuspid function
Preload is not critically dependant upon unimpeded pulmonary flow • Increases in PVR won’t significantly affect systemic circulation • Qp driving force is now SVC pressure • Qp must pass through two highly regulated vascular beds • Pulmonary and cerebral
Transpulmonary pressure gradient • Mean pulmonary arterial pressure – mean atrial pressure • Represents the driving force through the lungs • Low PVR allows for a low delta P • Which means lower SVC pressures
Pulmonary flow can be impaired by • High PVR • Increased atrial pressures • AV valve dysfunction • Ventricular diastolic dysfunction • A low transpulmonary gradient with a good CO means good things for sleep….
Post-Op issues – Ventilator Management • Excessive Paw • will limit systemic venous return via increased intrathoracic pressure • increase PVR, potentially decreasing pulmonary flow AND increases SVC pressure • Minimize iT, PIP and choose PEEP that allows for maintenance of FRC • Remember that Qp comes through the cerebral vascular bed… • So maneuvers like alkalosis and hyperventilation to decrease PVR may INCREASE cerebral vasc resistance, decreasing flow and further exacerbate hypoxemia