The Structure of the Atom
140 likes | 287 Vues
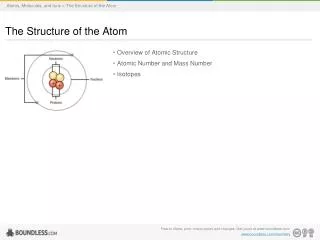
The Structure of the Atom. 4.2 Defining the Atom. Atoms are the smallest particle of an element that retains the properties of the element. How small are atoms? 50 MILLION atoms in a row would take up 1cm. Structure of Atom. Atoms are made up of protons (p+), neutrons (n), and electrons (e-)

The Structure of the Atom
E N D
Presentation Transcript
4.2 Defining the Atom • Atoms are the smallest particle of an element that retains the properties of the element. • How small are atoms? • 50 MILLION atoms in a row would take up 1cm
Structure of Atom • Atoms are made up of protons (p+), neutrons (n), and electrons (e-) • Protons = positive (+) charge • Electrons = negative (-) charge • Neutrons = neutral (no charge) • Atomic number = number of protons • Each element has ONLY ONE atomic number (# of protons)
Mass Number • Mass number = mass of the atom • Mass # = protons + neutrons Chemical Symbol Notation
Completing the Model of the Atom • Extremely dense nucleus with + protons & neutral neutrons. • Why don’t protons repel each other? • Most of the volume is empty space that electrons travel through.
4.3 How Atoms Differ • Atomic Number: number of protons (unique to each element) • For neutral atoms, #p+ = #e-
Ions are charged atoms. Lose e-’s and become positive. Gain e-’s and become negative. • F- • Ca2+
Ions & Isotopes • Ions: charged atoms (+/-) • (+) = less e- than p+ = cations • (-) = more e- than p+ = anions • Remember, you can’t change the number of p+ in an atom. You can change e- to get an ion • Isotopes: Atoms with same # of p+, but different # of neutrons • Remember, you can’t change the number of p+ in an atom. You can change n to get an isotope
How to Find Net Charge on Atom • Net charge = protons + (- # of electrons) • Ex) Carbon has a (-2) charge: 6 p+ + (-8e-) = -2 • Ex) Carbons has a (+3) charge: 6 p+ + (-3e-) = +3
Isotopes & Mass Number • Isotopes: same number protons, different number neutrons. • Ex. Potassium always has 19 p+ & 19 e- but it can have 20, 21, or 22 neutrons. • Mass Number: sum of p+ & n0.
IsotopesIn nature, most elements are found as mixtures of isotopes. Isotopes have essentially the same chemical & physical properties. Ex. Neon-22 Mass Number Atomic Number Ne 22 10
Give the # of p+, e-, and n0. SYMBOLS! • Carbon-12 • 6p+, 6e-, 6n0 • Carbon-13 • 6p+, 6e-, 7n0 • Carbon-14 • 6p+, 6e-, 8n0 12 6 13 6 14 6 C C C
Mass of Atoms • Atomic Mass Unit (amu): 1/12 the mass of a carbon-12 atom. • Protons: 1.007276 amu • Neutrons: 1.008665 amu • Electrons: 0.000549 amu • Atomic Mass: weighted average mass of the isotopes of that element. • The mass you see on the Periodic Table
Average Atomic Mass Chlorine • 76% chlorine-35 • 24% chlorine-37 • Atomic mass: 35.453 amu (mainly Cl-35) • (0.76 x 35) + (0.24 x 37) = 35.45 • Formula for finding average atomic mass: [% abundance X mass (amu)] + [% abundance X mass (amu)] = avg. mass