Chemical Equilibrium
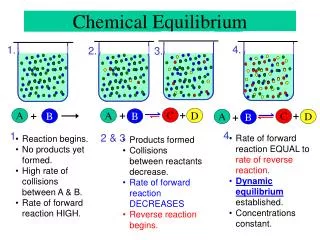
+. A. B. Chemical Equilibrium. 4. 1. 2. 3. +. +. +. C. A. D. B. C. +. A. D. B. 4. 1. 2 & 3. Rate of forward reaction EQUAL to rate of reverse reaction . Dynamic equilibrium established. Concentrations constant. Reaction begins. No products yet formed.


Chemical Equilibrium
E N D
Presentation Transcript
+ A B Chemical Equilibrium 4. 1. 2. 3. + + + C A D B C + A D B 4. 1. 2 & 3 • Rate of forward reaction EQUAL torate of reverse reaction. • Dynamic equilibrium established. • Concentrations constant. • Reaction begins. • No products yet formed. • High rate of collisions between A & B. • Rate of forward reaction HIGH. • Products formed • Collisions between reactants decrease. • Rate of forward reaction DECREASES • Reverse reaction begins.
EQUILIBRIM REACTIONS • Most reactions DO NOT go to completion. • Reactions that do not go to completion are REVERSIBLE. • Reversible reactions exist in a state of EQUILIBRIUM. • Equilibrium is reached when the rate of the forward reaction equals the rate of the reverse reaction. • The reaction proceeding from L R as the equation is written is known as the forward reaction. e.g. N2(g) + 3H2(g) 2NH3(g)
Chemical Equilibrium + + + + C A A D B B C + A D B …………… ……………… …………… ………….. …………………………………………… ……………… …………… ……………….
Chemical Equilibrium + + + + C A A D B B C + A D B DYNAMIC CHEMICAL EQUILIBRIUM Reactants Concentration Products time Forward Reaction Rate Reverse
Reactants Products time Equilibrium Position - Left • The equilibrium reaction does not mean the amounts of products and reactants are equal. • If theproducts react more easilythan the reactants but BOTH RATES are very slow. • We say the Equilibrium is shifted to the LEFT. • There will be higher concentration of reactants. + + C D A B Concentration Ratio of:Products Reactants Would be LESS than one. < 1
Equilibrium Position Middle • If the reactants and the productsboth have similar reaction rates. • The Equilibrium position will lie in the middle. • There will be the same concentration of reactants & Products. + + C D A B Reactants Concentration Ratio of: Products Reactants Would beEQUAL TO one. Products time = 1 Products Reactants = 1
Equilibrium Position - Right • If the reactants react ……… ………… than the products. • The Equilibrium is ………….. to the ………………. • There will be higher concentration of …………. + + C D A B …………… Ratio of: Products Reactants Would be………………. than ONE. Conc ……………. 1 time
Equilibrium Position - Right • If the reactants react more easily than the products. • The Equilibrium is shifted to the RIGHT. • There will be higher concentration of Products. + + C D A B Products Ratio of: Products Reactants Would beGREATER than one. Conc Reactants > 1 time
…………… Concentration …………… time Products Reactants Products Reactants > 1 < 1 Equilibrium Position SHIFT + To ………… + C D A B To ……… ………………. Reactants Concentration Concentration ………………. Products time time Products Reactants = 1
Products Concentration Reactants time Products Reactants Products Reactants > 1 < 1 Equilibrium Position SHIFT + + C D A B To right SHIFTED To Left Reactants Reactants Concentration Concentration Products Products time time Products Reactants = 1
Adding Reactants reactants Conc products + + time C A D B More …………………….added Reactant concentration …………………… Forward reaction rate ………………….. Reactant concentration ………………/Product concentration …………. Reverse reaction rate ………………………. New equilibrium established - ………….. ratio of PRODUCTS/REACTANTS - equilibrium shifted to …………………
Adding Reactants Reactant added reactants Equilibrium Shifts New equilibrium established Conc. products + + time C A D B More Reactant added Reactant concentration increases & Forward reaction rate increases Product concentration decreases & Reverse reaction rate decreases Reactant concentration decreases as reactants used up. Product concentration increases as new products formed. New equilibrium established - higher ratio of PRODUCTS/REACTANTS - equilibrium shifted to RIGHT
Adding Products New Equilibrium Eqm. Shifts Reactants Conc Products Product added + + time C A D B MorePRODUCTadded PRODUCTconcentration increases (Instantly - Reactantdecreases ) 3. REVERSE reaction rate increases (fast then slower) 4. PRODUCT concentration decreases, REACTANT concentration increases (fast then slower) 5. Forward reaction rate increases 6. New equilibrium established - lower ratio of PRODUCTS/REACTANTS - equilibrium shifted to LEFT.
Conditions of an equilibrium • At equilibrium both reactions continue to occur - the system is DYNAMIC. • The system is CLOSED – nothing added or removed. • The concentrations of reactants and of products remain constant. • Rate of the forward reaction equals rateof the back reaction. • Equilibrium can be obtained from either side.
H2(g) + I2(g) H2(g) + I2(g) 2HI(g) 2HI(g) Chemical Equilibrium Equilibrium is reached when the rate of the forward reaction equals the rate of the reverse reaction. (H = -13kJ/mol) The H value always refers to the forward reaction. 13 kJ of energy is liberated for every mole of HI formed. For the whole reaction: (H = -26kJ)
H2 + I2 2 HI 2 HI H2 + I2 H2 + I2 2 HI Chemical Equilibrium Reaction Rate Time
H2 + I2 2 HI 2 HI H2 + I2 H2 + I2 2 HI Chemical Equilibrium Reaction Rate Time
H2(g) + I2(g) 2HI(g) Kc = Equilibrium Constant At equilibrium the concentration of all sub- stances are constant at a fixed temperature. Each equilibrium has a constant e.g. for [H2] means conc. of ... N.B Kc is only determined by the concentrations of solutions and gases. Pure liquids & solids are not included in the equation – their concentrations are constant.
aA + bB cC + dD Equilibrium Constant In General: [C]c [D]d Kc = [A]a [B]b When Kc has a high value, there will be more PRODUCTS – (on the RIGHT) - we say the equilibrium lies to The RIGHT (vice versa for a low value).
Equilibrium Constant Calculations 1. Thebalanced equation must be known. • The concentration of a solid or liquid remainsconstant - these are not included in theequation. 3. The concentration of the solvent is constant and not included in the equation. 4. The value of Kc is given without units.
Kc Calculation Examples Write expressions for Kc for each of the following reactions: • C3H8(g) + 5O2(g) 3CO2(g) + 4H2O(g) • Ca(s) + 2H2O(ℓ) Ca(OH)2(aq) + H2(g) • AgNO3(aq) + NaCℓ(s) AgCℓ(s)+ NaNO3(aq) D. Na2CO3(s) + 2HCℓ (aq) 2NaCℓ(aq) + H2O(ℓ) + CO2(g)
Kc Calculation Examples [CO2]3[H2O]4 [C3H8] [O2 ]5 Write expressions for Kc for each of the following reactions: • C3H8(g) + 5O2(g) 3CO2(g) + 4H2O(g) • Ca(s) + 2H2O(ℓ) Ca(OH)2(aq) + H2(g) • AgNO3(aq) + NaCℓ(s) AgCℓ(s)+ NaNO3(aq) D. Na2CO3(s) + 2HCℓ (aq) 2NaCℓ(aq) + H2O(ℓ) + CO2(g) Kc = Kc = [Ca(OH)2][H2] [NaNO3] [AgNO3] Kc = [NaCl]2[CO2] [HCl]2 Kc =
Kc Calculation Examples 7 [NH3]2 [H2]3 [N2] (4/2)2 (1/2)3 (6/2) Kc = = 10.66 = 3H2 + N2 2NH3 Initial: 7mol 8mol 0mol Reacts: 6mol 2mol Eqm: 1mol 6mol 4mol [ ]: 1/2 6/2 4/2
Calculating K Given: N2(g) + 3H2(g) ↔ 2NH3(g) At equilibrium: [N2] = 0.30 M [H2] = 0.10 M [NH3] = 0.20 M
Calculating Concentrations at Equilibrium Methane gas reacts with water vapour to produce carbon monoxide gas and hydrogen gas according to this equation: CH4(g) + H2O(g) ↔ CO(g) + 3H2(g) At equilibrium: [CO] = 0.300 M [H2] = 0.800 M [CH4] = 0.400 M. If K is 5.67, calculate the concentration of water vapour Homework:complete the worksheet: Equilibrium Exercise 1
Carbon monoxide is a primary starting material in the synthesis of many organic compounds, including methanol, CH3OH(l). At 2000 0C, K is 6.4 x 10 −7 for the decomposition of carbon dioxide into carbon monoxide and oxygen. Calculate the concentrations of all entities at equilibrium if 0.250 mol of CO2(g) is placed in a closed container heated at 2000 0C.
If 0.50 mol N2O4(g) is placed in a 1.0 L closed container at 150 0C, what will be the concentrations of N2O4(g) and NO2(g) at equilibrium? (K=4.50) When hydrogen and iodine are placed in a closed container at 440 0C, they react to form hydrogen iodide. At this temperature, the equilibrium constant, K is 49.7. Determine the concentrations of all entities at equilibrium if 4.00 mol of hydrogen and 2.00 mol of iodine are placed in a 2.00 –L reaction vessel. H2(g) + I2(g) ↔ 2HI(g)
The following reaction has an equilibrium constant of 25.0 1100 K. H2(g) + I2(g) ↔ 2HI(g) If 2.00 mol of hydrogen gas, H2(g) and 3.00 mol of iodine gas, I2(g) are placed in a 1.00 L reaction vessel at 1100K, what is the equilibrium concentration of each gas?
Changing Equilibrium Conditions Predicting the effect Le Chatelier’s Principle If the conditions of an equilibrium system are changed, a process takes place which tends to oppose or cancel the effect of the change.
N2(g) + 3H2(g) 2NH3(g) (H < 0) Kc = Changing Equilibrium Conditions 2. The equilibrium constant • When the concentration of the N2 is increased, the ratio (Kc) will be smaller. • To restore the value of Kc and the equilibrium,more N2 will have to react with H2 • This will diminish the concentrations of reactants.
Kc = Adding Reactants N2(g) + 3H2(g) 2NH3(g) (H < 0) • When the concentration of the N2 is increased: • the concentration of reactants increases instantaneously • The ratio Kc (momentarily) decreases • Forward reaction rate increases and the concentrations of REACTANTS is LOWERED whilst the concentration of NH3 INCREASES. • The reverse reaction rate INCREASES as more NH3 forms. • A new equilibrium is established which has shifted to the RIGHT. • The value of the ratio Kc is restored to its original value Overall Kc UNCHANGED! Kc time
N2(g) + 3H2(g) 2NH3(g) (H < 0) Kc = Effect of Temperature • Increasing the temperature • will favour the ………………………. reaction. • In an EXOTHERMIC REACTION The ………………………reaction will be favoured • More ……………………..produced • the reaction shift to the ………….. Kc time The ratio (Kc) will therefore …………………………..
N2(g) + 3H2(g) 2NH3(g) (H < 0) Kc = Effect of Temperature • Increasing the temperature • will favour the ENDOTHERMIC reaction. • The REVERSE reaction will be favoured • More REACTANTS produced • the reaction shift to the LEFT. Kc time The ratio (Kc) will therefore DECREASE.
Kc = Effect of Pressure N2(g) + 3H2(g) 2NH3(g) (H < 0) • Increasing the PRESSURE • Equilibrium will shift to …………………..pressure. • Reaction will favour the side with the ……………………….number of MOLES of GAS – …………… • More ………….. produced. • Kc goes go ……………….(this eg)! Kc time The ratio (Kc) will therefore INCREASE.
Kc = Effect of Pressure N2(g) + 3H2(g) 2NH3(g) (H < 0) • Increasing the PRESSURE • Equilibrium will shift to REDUCE pressure. • Reaction will favour the side with the LEAST number of MOLES of GAS – RHS NH3. • More NH3 produced. • Kc goes go up (this eg)! Kc time The ratio (Kc) will therefore INCREASE.
Effect of a Catalyst Ea Ea E react E prod Potential Energy Potential Energy E prod E react • A CATALYST lowers the ACTIVATION ENERGY of the reaction by providing a different reaction pathway. • Activation Energy is lowered for ………………… FORWARD AND REVERSE REACTIONS. • BOTH rates are therefore increased by the ……………………and so the EQUILIBRIUM DOES ………………….. SHIFT!!!!
Effect of a Catalyst Ea Ea E react E prod Potential Energy Potential Energy E prod E react • A CATALYST lowers the ACTIVATION ENERGY of the reaction by providing a different reaction pathway. • Activation Energy is lowered for BOTH FORWARD AND REVERSE REACTIONS. • BOTH rates are therefore increased by the same amount and so the EQUILIBRIUM DOES NOT SHIFT!!!!
N2(g) + 3H2(g) 2NH3(g) (H < 0) Kc = Industrial Preparation • Favorable conditions: • High • High • LOW • LOW • In practice a ……………………. Temperature is used. Too ………………… will slow the reaction down. Kc time
N2(g) + 3H2(g) 2NH3(g) (H < 0) Kc = Industrial Preparation • Favourable conditions: • High [N2] & [H2] • High Pressure • LOW Temperature • LOW [NH3] • In practice a compromise temperature is used. Too low will slow the reaction down. Kc time
Changing Equilibrium Conditions Change Shift
Equilibrium Disturbance 2SO2(g) + O2(g) --> 2SO3(g) • Conc of O2 is increased • Temperature is increased • Pressure is increased
Equilibrium in solutions Heterogeneous equilibrium Depending on circumstances, a solution may be: • Under saturated – forward rate greater than reverse (salt dissolving) • Over saturated – reverse greater than forward – salt precipitates • Saturated– forward & reverse the same rate (equilibrium)
Equilibrium in solutions Heterogeneous equilibrium Depending on circumstances, a solution may be: • Under saturated – forward rate greater than reverse (salt dissolving) • Over saturated – reverse greater than forward – salt precipitates • Saturated– forward & reverse the same rate (equilibrium)
Solubility of salts • All nitrates are soluble • All alkali metal & ammonium salts are soluble • Chlorides, bromides & iodides are soluble - except Ag, Hg, Cu & Pb • Sulphates are soluble – except Pb, Ca, Ag &Hg • Carbonates, phosphates & sulphates of alkali metals & ammonium are soluble • Hydroxides of alkali metals, ammonium & barium are soluble • Sulphides of alkali metals, alkaline earth metals and ammonium are soluble
AB (s) A+ (aq) + B- (aq) Equilibrium in solutions The equation for the equilibrium reaction of a saturated salt solution can be represented as follows:
Ca(OH)2(s) Ca2+(aq) + 2 OH-(aq) Equilibrium in solutions The equilibrium constant is called the solubility productand is calculated as follows: Ksp = [Ca2+][OH-] 2
Equilibrium in solutions Temperature Change: Solubility curves show us that solubility's of most salts increase with increase in temperature.
Equilibrium in solutions Change in concentration : NaCl (s) Na+(aq) + Cl-(aq) Adding HCl to the above equilibrium, causes the equilibrium to shift to the left. NaCl is therefore precipitated until the equilibrium is restored. Disturbance of the equilibrium by increas- ing the concentration of one kind of ion is called the common ion effect.
Equilibrium in solutions Take note that the common ion effect is not restricted to solubility equilibria only. NH3(aq) + H2O(l) NH4+(aq) + OH-(aq) Adding a few drops of indicator and NH4Cl will show that the equilibrium will shift to the left - Explain.