Chemical Equilibrium
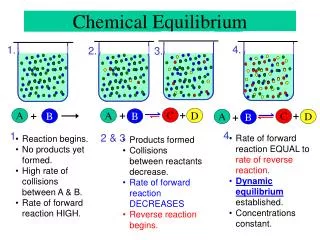
Chapter 15. Chemical Equilibrium. Dynamic equilibrium : a state when two opposing processes occur at equal rates. . Chemical Equilibrium: occurs when opposing reactions are proceeding at equal rates and the concentrations of products and reactants no longer change with time.

Chemical Equilibrium
E N D
Presentation Transcript
Chapter 15 Chemical Equilibrium
Dynamic equilibrium : • a state when two opposing processes occur at equal rates.
Chemical Equilibrium: occurs when opposing reactions are proceeding at equal rates and the concentrations of products and reactants no longer change with time. Ratef = Rater
Chemical Equilibrium For this to occur, the system must be closed, no products or reactants may escape the system.
For Reaction: AB Forward Reaction: AB Rate = kf[A] kf= forward rate constant Reverse reaction: BA Rate = kr[B] kr= reverse rate constant
As forward reaction occurs, • [A] decreases and forward rate slows. • [B] increases and the rate of reverse reaction increases. • Eventually the forward and reverse reactions reach the same rate. (Chemical Equilibrium)
kf[A]= kr[B] • Once equilibrium is established, the concentrations of A and B do not change.
Writing equilibrium expressions Keq = [products] [reactants] NOTE: [at equilibrium] Note: eq is the general subscript for an equilibrium constant. Your textbook uses Keq throughout. Please look at the equilibrium constants given on the AP exam. These are the same as Keq, but are more specific to a type of reaction (examples: gaseous reaction or acid base reaction).
Two common ways of describing equilibrium: Kc = Concentration of substances in the reaction are known. Kp = Units of partial pressure are used instead of concentration. *Kp is used when the reactants and products are in the gaseous state. *Solids and liquids are left out of equilibrium expressions because their concentrations do not change during chemical reactions.
Law of Mass Action: Expresses the relationship between concentrations of reactants and products at equilibrium. Example: aA + bB cC + dD Kc = [C]c[D]d [A]a[B]b
Kc = [C]c[D]d [A]a[B]b • The expression above is known as the equilibrium-constant expression. • Kcis the equilibrium constant. • Subscript c indicates concentration in molarity.
Write the equilibrium constant expressions for the following reaction in the forward direction. 2SO2(g) + O2(g) 2SO3(g)
Example:H2(g) + I2(g) 2HI(g) The equilibrium constant for this reaction at 400oC is 64. What is the equilibrium constant value for the reverse reaction? Kc fwd = [HI]2 [I2][H2] 64 = [x]2 x = 8
H2(g) + I2(g) 2HI(g) Kc rev = [H2][I2] [HI]2 Kcrev = [1][1] [x]2 Kcrev = 1 [x]2 Reverse = .016
The Haber Process N2(g) + 3H2(g) 2NH3(g) • This process for making ammonia is done at high temperature and pressure. • When complete all 3 components are present in the closed tank.
Regardless of the starting concentrations, at equilibrium the relative concentrations of the 3 gases are the same.
Equilibrium constant for Haber Process • The equilibrium-constant depends only on the stoichiometry of the reaction, not on its mechanism (process by which it occurs).
The following equilibrium process has been studied at 230ºC: 2NO(g)+ O2(g)2NO2(g) In one experiment theconcentration of the reacting species at equilibrium are found to be [NO]=0.0542M [O2]=0.127M [NO2]=15.5M. Calculate Kc of the reaction at this temperature.
Kc in terms of pressure Example: aA + bB cC + dD PA is partial pressure of A in atm, PB is the partial pressure of B, etc.
Values for Kc and Kp are usually different. • It is possible to calculate one from the other. • n is the change in the number of moles of gas. Δn = nproducts - nreactants
N2O4(g) 2NO2(g) • n = 2-1 = 1 • For the above reaction, Kp=Kc(RT)
For the equilibrium 2SO3(g) 2SO2(g) + O2(g) at temperature 1000K, Kc has a value 4.08 x 10-3. Calculate the value for Kp. Kp = 4.08 x 10-3 (.0821 x 1000)1 0.335
A 3:1 starting mixture of hydrogen, H2, and Nitrogen, N2 comes to equilibrium at 500 °C. The mixture at equilibrium is 3.506% NH3, 96.143% N2, and 0.3506% H2 by volume. The total pressure in the reaction vessel was 50.0 atm. What is the value of Kp and Kc for the reaction? N2 + 3H2 2NH3 Hint: Use Dalton’s Law of partial pressures to determine the partial pressure of each component in the reaction. PA = XA PT Since we know we have 100%, Partial Pressure = (% of substance /100 X total pressure)
P NH3 = 0.03506 x 50.0 atm = 1.75 atm P N2 = 0.96143 x 50.0 atm = 48.1 atm P H2 = 0.003506 x 50.0 atm = 0.175 atm (P NH3)2 (1.75)2 Kp = = =11.9 (P N2) (P H2)3 (48.1) (0.175)3 Δn ( 1 RT ) Kc = Kp -2 ( 1 0.0821 x 773 ) = 4.79 x 104 Kc = Kp
Magnitude of Equilibrium constants • When Kc is very large the numerator is much larger than the denominator. • The equilibrium lies to the right, or to the product side of the equation. K =[products] [reactants]
When the equilibrium constant is very small the equilibrium lies to the left, or the reactant side. • K>>1 Products favored. • K<<1 Reactants favored.
Direction of equation and K • Equilibrium can be approached from either direction. • The equilibrium constant for an equation written in one direction is the reciprocal of the constant for the other direction.
Direction of equation and K N2O4(g) 2NO2(g) For forward Rx: For reverse Rx:
Heterogenous Equilibria • Substances that are in equilibria and are in different phases. CaCO3(s) CaO(s)+ CO2(gas) Equilibrium constant
The concentration of a pure solid or liquid is a constant. • If a pure solid or pure liquid are involved in a heterogeneous equilibrium, its concentration is not included in the equilibrium-constant expression for the Rx.
This equation shows that at a given temperature, an equilibrium between CaO,CaCO3, & CO2 will always lead to the same concentration of CO2. • Pure solids or pure liquids must be present and participate for equilibrium to be established.
Write the equilibrium expression Kc and Kp for the following reaction: 3Fe(s)+ 4H2O(g) Fe3O4(s) + 4H2(g)
Calculating Equilibrium Constant “When all Equilibrium Concentrations are not known.”
Calculating Equilibrium Constants • Equilibrium concentrations are often unknown. • If one equilibrium concentration is known, we can use stoichiometry to find the equilibrium concentration of the other species.
Equilibrium Concentrations Initial concentration Change in concentration Equilibrium concentration
Handout Procedure for calculating equilibrium concentrations. H2(g) + I2(g) 2HI(g) 1) First, we design our table so there is a column for each component in the equation, and rows for start, Δ, and finish.
H2(g) + I2(g) 2HI(g) • Next, we fill in what we know. • *Since HI is a product, it is 0 at the start of the reaction. • *We also know that at equilibrium, 3.50 mol of HI exist.
H2(g) + I2(g) 2HI(g) 3) We now use stoichiometry to fill in the rest of the table.
H2(g) + I2(g) 2HI(g) 3.50 mol HI 1 mol H2 = 1.75 mol H2 2 mol HI *It is negative because it is being used up in the reaction.
H2(g) + I2(g) 2HI(g) 3.50 mol HI 1 mol I2 = 1.75 mol I2 2 mol HI *Now we can fill in the “finish” column.
H2(g) + I2(g) 2HI(g) The quantities in the finished row are used to calculate Kc. (HI)2 (3.50)2 = Kc = =196 (H2) (I2) (0.25) (0.25)
You try this one! (On back of handout) Nitric oxide gas, NO, and oxygen gas, O2, react to form the poisonous gas nitrogen dioxide, NO2, in the reaction shown below: 2NO(g) + O2(g) 2NO2(g) 10.0 moles of NO and 6.00 moles of O2 are placed into an evacuated 1.00 L vessel, where they begin to react. At equilibrium, there are 8.80 moles of NO2 present. Calculate the value of Kc, assuming that the temperature remains constant throughout the reaction.
10.0 moles of NO and 6.00 moles of O2 are placed into an evacuated 1.00 L vessel, where they begin to react. At equilibrium, there are 8.80 moles of NO2 present. Calculate the value of Kc, assuming that the temperature remains constant throughout the reaction. 2NO(g) + O2(g) 2NO2(g)
When 3.0 mol of I2 and 4.0 mol of Br2 are placed in a 2.0 L reactor at 150oC, the following reaction occurs until equilibrium is reached: I2(g) + Br2(g) 2IBr(g) Chemical analysis then shows that the reactor contains 3.2 mol of IBr. What is the value of the equilibrium constant Kc for the reaction? Consider M since we have a 2.0 L vessel.
3.0 mol of I2 and 4.0 mol of Br2 are placed in a 2.0 L reactor at 150oC. Chemical analysis then shows that the reactor contains 3.2 mol of IBr. What is the value of the equilibrium constant Kc for the reaction? I2(g) + Br2(g) 2IBr(g)
If you are dealing with the pressures of gases at equilibrium, you will be given: moles, temperature, and volume. is the key to calculating the pressure of each gas at equilibrium. Stoichiometric equivalents may also be used. PV = nRT
Example: Handout 2SO3(g) 2SO2(g) + O2(g) Initially, 0.01219 moles of SO3 gas in a 2.0 L vessel are heated to a temperature of 1000K where it reaches equilibrium. At equilibrium the vessel is found to contain 0.00487 moles of SO3 gas. A) Calculate the equilibrium partial pressures of SO2(g) and O2(g). B) Calculate Keq.
1) We must first calculate the pressures from the information given in the problem. PV = nRT P = nRT/V P = .01219(0.8206)(1000) / (2.0) = 0.500 atm This is the initial pressure of SO3 gas. 2SO3(g) 2SO2(g) + O2(g)
PV = nRT P = nRT/V P = .00487(0.8206)(1000) / (2.0) = 0.200 atm This is the pressure of SO3 gas at equilibrium. 2SO3(g) 2SO2(g) + O2(g)