Chemical Equilibrium
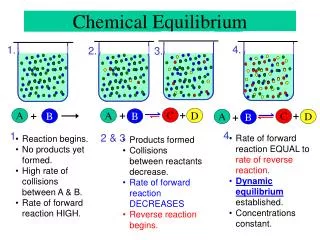
Chemical Equilibrium. Recognizing Rxn Equilibrium. 1. The system is closed. Opposite rxns occur at the same rate. Equilibrium is reached by either starting w/ reactants or products. Temp is constant.

Chemical Equilibrium
E N D
Presentation Transcript
Recognizing Rxn Equilibrium 1. The system is closed. • Opposite rxns occur at the same rate. • Equilibrium is reached by either starting w/ reactants or products. • Temp is constant. **Radioactive tracers can prove th rxn proceeds even w/ no net observable changes: dynamic equilibrium Equilibrium does NOT mean conc of P+R are equal, but th RATES ARE EQUAL!
Equilibrium Constant, Keq • A <=> B • Once equilibrium is reached, the ratio of partial pressures of A and B is constant. • Law of Mass Action • Shows the relationship between the concs (expressed as partial pressures for gases & as molarities for solutions) of the R + P present at equilibrium in any reaction.
Equilibrium Constant, Keq-cont • aA + bB <=> cC + dD • Equilibrium-constant Expression: • Keq = [C]c[D]d [A]a[B]b • Temp dependent • Dimensionless: no units
Magnitude of Keq • Keq can be very large or very small • CO(g) + Cl2(g) <=> COCl2(g) • Keq = PCOCl2 = 1.49 x 108 PCOPCl2 For Keq to be so large >> 1, the numerator, PCOCl2, must be large: The equilibrium lies to the right (toward the P) Keq <<1: equilibrium lies to the left (reactants dominate)
Direction of Chemical Equilibrium & Keq • Since equilibrium can be approached fr either the P or R, wh way we write the equation is arbitrary N2O4 <=> 2NO2 • Keq = (PNO2)2 = 6.46 P N2O4 2NO2 <=>N2O4 • Keq = P N2O4 = 0.155 (PNO2)2 The Keq expression for a rxn written in one direction is the reciprocal of the one for the rxn written in the reverse direction.
Other Ways to Manipulate Eq & Keq Values • When using Hess’s Law to obtain a net equation by adding equations & canceling like terms: • The Keq of a rxn in the reverse direction is the inverse of Keq of the forward rxn. • The Keq of a rxn th has been multiplied by a number is the Keq raised to a power equal to th number. • The Keq for a net rxn made of 2 or more steps is the product of the Keq for the individual steps.
Heterogeneous Equilibria • Homogeneous equilibria -involve subs all in the same phase • Heterogeneous equilibria - subs are in diff phases • Partial pressures of gases are used in Keq expression. • Molar conc of dissolved species are used. • Pure solids, pure liquids, and solvents are NOT included in Keq expression. * these subs must be present for equilibrium
Calculating Keq • If we know the equil conc of at least one species in a rxn, we can use stoichiometry of the rxn to determine the conc of the other species at equilibrium.
Calculating Keq-cont • Tabulate the known initial & equilibrium concs of all species in the Keq expression. • For those where initial & equil conc are known, calc change in conc. • Use stoich of rxn to calc changes in conc for all other species. • Fr initial & change in conc, calc the equil conc.
Applications of Keq • Allows us to predict: • 1) the direction in wh the rxn will proceed to achieve equilibrium • 2) calculate the conc of R+P when equilibrium has been reached
Applications of Keq-cont. • Predicting the Direction of Rxn Q - rxn quotient- calc by substituting in partial press or conc into Keq expression Q = Keq if sy is at equilibrium Q > Keq- reaction proceeds to the left to reach equil Q< Keq - reaction proceeds to the right
Applications of Keq-cont • Calculating Equilibrium Constants - Again use the known initial & equil conc - Set up a ICE table - Calc unknown equil conc
Le Chatlier’s Principle • If a sy at equilibrium is disturbed by a change in temp, press, or the conc of comps, the sy will shift the eq position to counteract the effect of the disturbance.
Le Chatlier’s Principle-cont • Change in R or P Conc • Adding a R or P disturbs equil: • Equil will re-establish by consuming the added R or P • Removing R or P • Equi will re-establish by forming more sub • N2(g) + 2H2(g) <=> 2NH3(g) • Add N2 or H2: shifts right • Add NH3: shifts left • Remove N2 or H2: shifts left • Remove NH3: shifts right
Le Chatlier’s Principle-cont • Effects of Volume & Press Change • ONLY effect gas phase! • A decrease in vol will ^ press • Eq will shift to decrease the press • Eq will shift to the side with the least number of gas particles N2O4(g) <=> 2NO2(g) ^ press will shift to the left
Le Chatlier’s Principle-cont • Effect of Temp Change • Temp will change the Keq • Endothermic: R + heat <=> P • ^ the temp will favor the endothermic rxn • ^ T = ^ Keq • Exothermic : R <=> P + heat • Decreasing the temp will favor the exo rxn • Increasing T = decrease in Keq
Le Chatlier’s Principle-cont • The Effect of Catalysts • Lowers the Ea for both the forward & reverse rxns • Increases the rate at wh equil is achieved, it will not shift the equilibrium or change the equilibrium mixture.
Haber Process • N2(g) + 2H2(g) <=> 2NH3(g) • High conc of H2(g) + N2(g)maintained. • NH3(g)(g) th forms is removed. • High temp to ^ the forward rxn. • 300-600˚C • Catalyst used to ^ rxn rate (est equilibrium faster) • High press used: 200-600 atm