Chemical Equilibrium
320 likes | 706 Vues
Chemical Equilibrium. Foundation of equilibrium Expressing equilibrium: Equilibrium constants Upsetting equilibrium – Le Chatelier. Learning objectives. Write equilibrium constant expressions for both solutions and gas phase reactions Deduce relationship between K c and K p

Chemical Equilibrium
E N D
Presentation Transcript
Chemical Equilibrium Foundation of equilibrium Expressing equilibrium: Equilibrium constants Upsetting equilibrium – Le Chatelier
Learning objectives • Write equilibrium constant expressions for both solutions and gas phase reactions • Deduce relationship between Kc and Kp • Use reaction quotients to predict direction of reaction • Apply LeChatelier’s principle to predict consequences of changing equilibrium conditions
Equilibrium • Chemical equilibrium is: The state reached when the concentrations of reactants and products remain constant over time • Equilibrium is evident in physical processes: vapour pressure over a liquid, solid and liquid coexisting at the freezing point
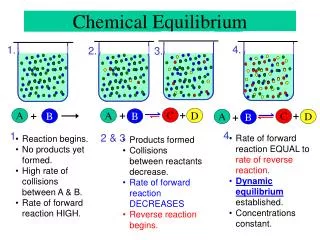
Equilibrium in a chemical change • Not all reactions proceed to completion – reactants completely converted into products • As reactants convert into products, the products themselves convert back into reactants • As reactant concentration declines, the forward rate decreases • As product concentration increases, the backward rate increases
Dynamic equilibrium • In the reaction The same final concentrations of NO2 and N2O4 are obtained regardless of the initial conditions – pure NO2 or pure N2O4 • The reactions don’t stop, the processes become equal in both directions
Reactants and products no more • In an equilibrium mixture, there is a constant, dynamic cycling of materials and the identities of reactant and product are lost • By convention, the substance(s) used initially are called the reactants
Rate perspective • As the concentration of reactant (product) decreases, the rate of disappearance also decreases • The equilibrium point is reached when the rates are equal
Equilibrium constant • Reactions usually take place in solution or in the gas phase. For solution reactions, the equilibrium constant Kc is used, where concentrations are in mol/L • For the equilibrium aA +bB = cC + dD Concentration M Products Coefficients from equation Reactants
Notes about Kc • Kc is a constant at a given temperature, regardless of the concentrations of the reactants used • Substances may be gases or solutions • Kc has no units: each concentration is considered as a ratio to the standard state (1 M)
Equilibrium constant Kp • For reactions in the gas phase, the partial pressure can be used instead of molarity • Kp is unitless because the ratio with respect to the standard state (1 atm) is used
Relationship between Kc and Kp PAV = nART so PA = nA/V●RT = [A]RT Δn = (c+d)-(a+b)
Heterogeneous equilibria • Reactions often involve solid or liquid phases in addition to gas and solution • Concentrations of liquids and solids are constants – independent of the amount • Since they are constant they become folded into Kc
Solids and liquids are ignored CaCO3(s) = CaO(s) + CO2(g) • Include only gas or solution entities in Kc Solid concentrations are constant
Using equilibrium constants • The value of Kc or Kp indicates the extent of a reaction • Generally: • Kc > 103, reaction goes to completion • Kc < 10-3, reaction does not proceed • 103 > Kc > 10-3 reactants and products all present
Reaction Quotient • Reaction Quotient is the instanteous value obtained for Kc for a combination of reactants and products not yet at equilibrium • It can be used to predict the direction the reaction will take from that set of conditions
Kc and Qc • In the reaction • A mixture made up with [H2] = 0.1 M, [I2] = 0.2 M and [HI] = 0.4 M • Kc > Qc so reaction will go towards HI
Predicting reactions with Qc • Qc < Kc, reaction goes towards products • Qc > Kc, reaction goes towards reactants • Qc = Kc, reaction is at equilibrium
Calculations with Kc • Simple calculations: Kc and all equilibrium concentrations but one are given. Use Kc to calculate concentration of unknown • Complex calculations: Kc and initial concentrations are given, calculate equilibrium concentrations
I.C.E. – It’s pretty cool • The ICE man cometh: it is widely used in equilibrium problems • Given Kc = 57 and initial concentrations of [H2] = [I2] = 0.1 M, find equilibrium concentrations of H2, I2 and HI
Solve for x • X = 0.136 > 0.1 so is physically unreasonable • X =0.0791: [H2] = [I2] = 0.0209 M; [HI] = 0.158 M
Upsetting the applecart • Conditions of reactions must often be manipulated to optimize yield of products • Concentrations of reactants or products • Temperature • Pressure and volume • Le Chatelier’s Principle: If a stress is applied to a reaction mixture at equilibrium, the system adjusts to relieve the stress
The Haber process • One of the most important industrial processes is the synthesis of NH3 from the elements: N2(g) + 3H2(g) = 2NH3(g) • At 700 K, Kc = 0.29 • Altering reactant concentrations: • Increasing [reactant]: reactants → products • Increasing [product]: products → reactants
Effects of pressure • Pressure is only important if there is an overall change in the number of gas moles N2(g) + 3H2(g) = 2NH3(g) • In the Haber process there are 4 moles of reactants vs 2 moles of products • Increasing pressure: converts reactants to products (fewer moles) • Decreasing pressure: converts products to reactants (more moles)
Effects of temperature • Kc depends on temperature. Increasing or decreasing temperature will cause mixture to adjust to new value of Kc. • Treat the ΔH of a reaction as a reactant/product • Raising T causes heat input into reaction • Lowering T causes withdrawal of heat
The Haber process as Function of T • The process is exothermic: heat is a product • Raising T puts heat into reaction: • Equilibrium adjusts to reduce heat output – moves towards reactants • Lowering T removes heat: • Equilibrium adjusts to increase heat output – moves towards products • Endothermic reactions will show the opposite T- dependence N2(g) + 3H2(g) = 2NH3(g) + 92.2 kJ
Catalysts and equilibrium • Equilibrium depends on the initial and final states • Catalyst lowers the transition state • Equilibrium is unaffected by addition of a catalyst
From the rate perspective • The catalyst increases the forward reaction rate by lowering the energy barrier • The rate of the backward reaction is lowered by the same amount
Linking rate equations with Kc • For the general reaction: A + B = C + D • Assume single bimolecular steps • Rate of forward reaction = kf[A][B] • Rate of backward reaction = kr[C][D] • At equilibrium