Chemical Equilibrium
Chemical Equilibrium. Be sure you download this powerpoint from the website. . The Concept of Chemical Equilibrium. Chemical equilibrium exists when the rates of the forward and reverse reactions are equal and the concentrations of the reactants and products remain constant.


Chemical Equilibrium
E N D
Presentation Transcript
Chemical Equilibrium Be sure you download this powerpoint from the website.
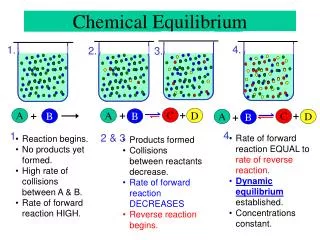
The Concept of Chemical Equilibrium • Chemical equilibrium exists when the rates of the forward and reverse reactions are equal and the concentrations of the reactants and products remain constant. • In other words… + Breactant C+ Dproduct • Occurs in a closed system, and no observable changes are detected.
Chemical Equilibrium • Chemical Equilibrium-involves different substances as reactants • Physical equilibrium- involves equilibrium between the phase changes of the same substance. • Example: physical equilibrium of water in a closed system:
Physical equilibrium • Physical equilibrium of water in a closed system: • How would this look in an open system? • At a high temperature (above freezing)? • At a low temp (below freezing)? • At a REALLY low temp (really below freezing)?
The Equilibrium Constant (K) • AP exam questions will either give you K to calculate a concentration of a substance, or give you concentrations to calculate K. • K is the mathematical expression of the law of mass action • Law of mass action • For a reversible reaction at equilibrium and a constant temperature, a certain ratio of reactant and product concentrations has a constant value, K.
The Equilibrium Constant (K) • General Equation: K= Where: aA + bB+ dD Mole coefficients are represented by a,b,c,d. Concentrations are expressed in Molarity (n/L)
The Equilibrium Constant (K) • General Equation: K= • Explained in English: the equilibrium constant is equal to the concentration of products divided by the concentration of reactants. • Concentrations are multiplied to the exponent of their mole coefficients (a, b, c, d).
The Equilibrium Constant (K) • General Equation: K= • When K > 1, there are more products than reactants. • When K<1, there are more reactants than products. • When K=1, they are equal.
The Equilibrium Constant (K) • For a homogenous equilibrium-all reacting species are in the same phase. • Example: N2O4(g)2NO2(g) Dinitrogentetraoxide gas “dissociates” in to Nitrogen dioxide gas. A reversible reaction that exists in equilibrium in a closed system.
The Equilibrium Constant (K) • In a homogenous equilibrium: N2O4(g)2NO2(g) = Subscript K indicates that concentrations are expressed in molarity. When concentrations are expressed as partial pressures, concentration is represented by Kp.
The Equilibrium Constant (K) • When concentrations are expressed as partial pressures, concentration is represented by Kp. N2O4(g)2NO2(g) Our equation then becomes: Kp = Where Ppartial= Xi*PT
The Equilibrium Constant (K) • Remember that is our mole fraction for a heterogeneous mixture of gases: = • And Total pressure is the sum of the individual partial pressures: Ptotal=∑P1∞ And… pv=nRT
The Equilibrium Constant (K) • Therefore: N2O4(g)2NO2(g) • Kp = • Kp= =(RT)b-a Where: b-a = ∆n A helpful hint is to read the equations to yourself as if you were translating them from a foreign language to English.
The Equilibrium Constant (K) • Since: Kp= =(RT)b-a • And because: b-a = ∆n Then: Kp=(RT)∆n =Kc(RT) ∆n because= And : Kp= Kc (0.0821*T) ∆n
Homework… • Read ReadReadReadReadReadReadReadReadReadReadReadReadReadReadReadReadchapter on Gases and on Chemical Equilibrium!!!!!! • You will have a quiz tomorrow on gas stoichiometry. I will assign you homework on equilibrium tomorrow. You will have a quiz on equilibrium Thursday.
Chemical Equilibrium Part II
Heterogeneous Equilibrium • Heterogeneous Equilibrium-results when reactants and products are in different phases. • Example: calcium carbonate is heated in a closed system, the following equilibrium is attained: CaCO3(s)CaO(s) + CO2(g) The presence of two solids and one gas represents three separate (not different) phases.
Heterogeneous Equilibrium CaCO3(s)CaO(s) + CO2(g) • The concentration of a solid is dependent on its density, not how much of the substance we have. Therefore; We can use our conversion factors to calculate concentration. • For example, Copper (Cu) has density of 8.96 g/cm3 Therefore, [Cu] == .141mol/cm3 Or 141 mol/L
Heterogeneous Equilibrium CaCO3(s)CaO(s) + CO2(g) The density of CaCO3(s)is 2.71 g/cm3 The density of CaO(s) is 3.35 g/cm3 And since BY DEFINITION the concentration of a solid is itself a constant AT CONSTANT TEMPERATURE and pressure; K’c=, since they are constant: K’c=Kc=[CO2]…wth…? CO2 is the only product that is a gas.
Heterogeneous Equilibrium • Example: • (NH4)2Se(s) 2NH3(g) + H2Se(g) • Remember that Kc= • so: Kc=? • And since the concentration of a solid is constant, we disregard it, therefore: Kc= If the concentrations are given in molarity…if the concentrations are given in partial pressures, things get real…
Heterogeneous Equilibrium • If the concentrations are given in partial pressures (NH4)2Se(s)2NH3(g) + H2Se(g) Kp=P From: Kc= Since the solid has no partial pressure, we ignore it.
Just in case you were wondering why… • If you were to calculate the pressure of a solid using the ideal gas equation PV=nRT where [A]=n/v • Lets use Copper again… • Copper (Cu) has density of 8.96 g/cm3 • A concentration of 141 mol/L at STP • Therefore; P/RT=n/v, assume STP, and one liter of substance. P==3,160 atm This means we would need 3160atm to convert a gas into a solid. In other words, you cant use the Ideal gas equation with solids…
Multiple Equilibria • The reactions we have considered are relatively simple, a more complicated situation is one in which the product molecules in one equilibrium system are in coved in a second process. aA + bB↔cC+ dD cC + dD ↔ eE + fF
Multiple Equilibria • When: aA+ bB↔cC + dD cC + dD ↔ eE + fF • Then: • Kc’ (for the first reaction)= • Kc’’ (for the second reaction)=
Multiple Equilibria • When: aA+ bB ↔ cC+ dD cC + dD ↔ eE + fF • (don’t write this down because it is confusing) The overall reaction is given by the sum of the two reactions: aA + bB ↔ cC+ dDK’c cC + dD ↔ eE + fFK’’c aA + bB ↔ eE + fFKc
Multiple Equilibria • The overall reaction is given by the sum of the two reactions: aA+ bB ↔ cC + dDK’c cC+ dD ↔ eE + fFK’’c aA+ bB ↔ eE + fFKc • Then the equilibrium constant for the overall reaction = Kc=
Multiple Equilibria • Therefore: if a reaction can be expressed as the sum of two or more reactions, the equilibrium constant for the overall reaction is given by the product of the equilibrium constants of the individual reactions
Multiple Equilibria • In other words: • Kc for multiple reactions: Kc = (Kc’) x (Kc’’) SO: Kc = Kc’ x Kc’’
The relationship between equilibrium and rate of reaction (kinetics) • For equilibrium reactions, we have both forward and reverse reactions, therefore; aA + bB ↔ cC + dD • Where Kfis our forward equilibrium constant, and Kris our reverse constant. Ratef = Rater Kf Kr
The relationship between equilibrium and rate of reaction (kinetics) • If, Ratef = Rater • And Ratef = kf[A]a[B]b (reactants) • And Rater = kr[C]c[D]d (products) • Then; =kc =
Next Week • We will cover • The reaction quotient (Q) • Le Chateliers Principle. • Please read sections 13.5, 13.6, and 13.7 in your book.
Homeworkdue Thursday. • Pgs 650-651, 17-35 odd #’s • Pgs 651-653, #’s 37-45 odd, 51, 55, 57.
Chemical Equilibrium Part III
The reaction quotient (Q) • For reactions that have not reached equilibrium, we obtain the reaction quotient (Q) instead of the equilibrium constant (K). • Simply put, the reaction quotient uses the initial concentrations of reactants and products in the same equation as K. • You then compare Q to K to determine whether or not the reaction is at equilibrium. • Simple enough.
The reaction quotient (Q) • Therefore, Q= When we are given K and asked to find Q: Q<K, more reactants than products Q=K, system is at equilibrium Q>K, more products than reactants. The system must balance itself to get to equilibrium.
Example • Consider the following reaction: N2(g) + 3H2(g) ↔2NH3(g) • at the start of the reaction there are .249 mol of N2, 3.21 x 10-2mol H2, and 6.42 x 10-4mol NH3 in a 3.5L reaction vessel at 375°C, If the equilibrium constant (K) is 1.2, decide whether the system is in equilibrium. If it is not, predict which way the net reaction will occur.
Example N2(g) + 3H2(g) ↔2NH3(g) • First calculate the concentrations (n/v) • Then calculate Q, Q= • compare Q to K(given) • predict direction of reaction to reach equilibrium
Example • Q=.611 • Q < K • Therefore, there are more reactants than products, and the reaction must proceed from left to right to reach equilibrium.
LeChatlier’s Principle • if an external stress is applied to a system at equilibrium, the system will adjust in such a way that the stress will is partially offset as the system reaches a new equilibrium position. • Stresses include: change in Temp, volume, pressure, or a change in moles.
LeChatlier’s Principle A classic example from Biology: • Your body is in a state of dynamic equilibrium with your environment. When you dramatically change environments, you body feels the effects and works to return to equilibrium. This is a process called “acclimatization”.
A classic example from Biology • The combination of oxygen with a hemoglobin molecule produces the polymer Oxyhemoglobin which transports oxygen to body tissues. Hb(aq) + O2 ↔ HbO2(aq)
Hb(aq) + O2 ↔ HbO2(aq) • At higher altitudes the concentration of oxygen decreases, thus leading to a state of disequilibrium. This is known as altitude sickness and can be fatal. • Yuppies call this “the height hangover”.
So according to LeChatlier: Hb(aq) + O2 ↔ HbO2(aq) • A decrease in oxygen would shift the equilibrium from left to right. In other words, the left side of the arrow above would be bigger that the left; and Q >K. This would mean that there is more HbO2 than what the environment realistically allows as a result of a smaller O2 concentration. • Your body (as the system) reacts by creating more Hb to capture more oxygen. This process takes weeks, even years.
Tomorrow • We will wrap up this chapter • Calculating equilibrium concentrations • Addressing system stresses Use the rest of the period to work on your homework.
Chemical Equilibrium Part IV
Calculating Equilibrium Concentrations • If we know the equilibrium constant K for a particular reaction, we can calculate the equilibrium concentrations from the initial concentrations Using the ICE method • Initial, Change, Equilibrium • We do this by solving algebraically.
Calculating Equilibrium Concentrations • Consider the following reaction of cis-stilbene and tran-stilbene, two isomers that exist in equilibrium in a closed system.
Kc for this system at 200° C is 24.0. if the initial concentration of cis-stilbene is .850 mol/L, and no trans-stilbene, what is the concentrations for both substances at equilibrium? • Solve algebraically using the ICE method
Cis-stilbene ↔ trans-stilbene Initial (M): .850 0 Change(M): -x +x Equilibrium (M): (.850 – x) x So: Kc== Solve for x