Basics of Biochemistry: Carbohydrates, Lipids, Proteins, and Nucleic Acids
E N D
Presentation Transcript
BIOCHEMISTRY • The chemistry of the carbon atom • Versatility of the carbon atom • Bonds readily to itself, forms chains, rings, single and double bonds • Hydrocarbons • Isomers • Polymers
I. Carbohydrates • Sugars and their polymers • CHO in a 1:2:1 ratio • An –OH group is attached to each C except one which is double bonded to Oxygen • Most common: • Triose 3C glyceraldehyde • Pentose 5C ribose • Hexose 6C glucose
A. Monosaccharides • Simple sugars • C6H12O6 – glucose, fructose, galactose • Arrangement of individual atoms makes them different compounds • Glucose – hexose – building block for more complex sugars • Fructose – fruit sugar • Galactose – milk sugar
B. Disaccharides • 2 monosaccharides joined by a glycosidic linkage through dehydration synthesis • Maltose = glucose + glucose (brewing beer) • Lactose = glucose + galactose (milk) • Sucrose = glucose + fructose (table sugar)
C. Polysaccharides • Polymer of a few hundred or thousand monosaccharides • 2 important functions: • 1. Energy storage (starch and glycogen) • 2. Structural support (cellulose and chitin) Starch = alpha linkage Cellulose = beta linkage
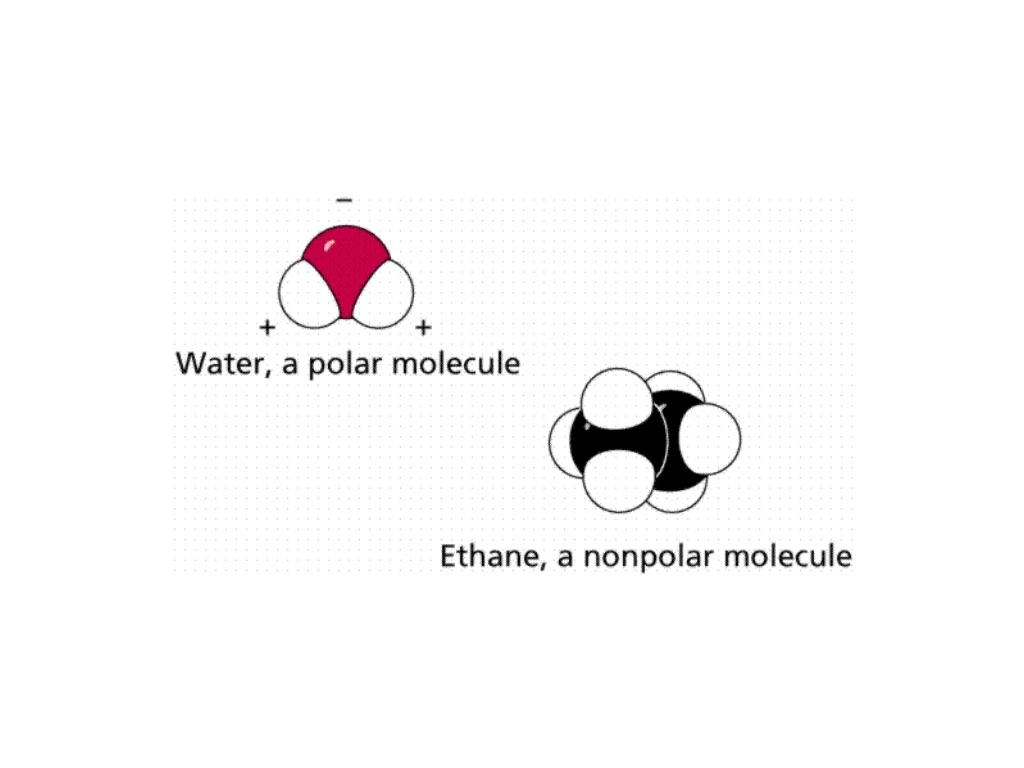
II. LIPIDS • Waxy or oily organic compounds • Nonpolar; CHO in no particular ratio • Fats, phospholipids, steroids • Functions: • Energy storage • Bio membranes • Chemical messengers • Insulation • Cushion vital organs
A. Fats • Glycerol and 3 fatty acids • Simple fat = triglyceride • Properties depend on the fatty acids that it is composed of • Saturated vs. unsaturated fatty acids
B. Phospholipids • Glycerol, fatty acids (2) and a phosphate group (negatively charged) • AMPHIPATHIC
C. Steroids • Lipids with 4 fused carbon rings with various functional groups attached • Amphipaathic • Cholesterol, hormones
III. PROTEINS • Polymers of amino acids • Amino acids are the monomers
III. PROTEINS cont’d • Amino acids can form covalent bonds with each other • 20 AA in nature – each has a different “R” group • R groups can be • Polar • Nonpolar • special
III. PROTEINSFormation • Two amino acids form a peptide bond through dehydration synthesis • Peptide bond formed between acid group of one amino acid and and amino group of another • 40 – 1,000 AA in MOST cellular proteins
PROTEINSStructure • Primary – linear sequence of amino acids • Secondary – folding due to H bonds (weak forces) (alpha helix, beta pleated sheet) • Tertiary – more folding of helix due to hydrophobic/hydrophilic interactions of R groups • Quaternary – two or more chains held together by weak forces
PROTEINSFunctions • Membrane structure (integral and peripheral) • Enzymes (biological catalysts) • Motion (muscle contraction) • Energy (last resort) • Transport (hemoglobin) • Hormones (cellular control) • Regulation of gene function • Protection (immune system)
III. PROTEINS cont’d • Denaturing occurs when temperature or pH changes or through treatment with urea • 3D structure is destroyed – no function • Primary structure is still intact; peptide bonds are strong covalent bonds that cannot be broken by change in pH, temp, etc
IV. NUCLEIC ACIDS • DNA and RNA, molecules which contain directions to make proteins; determine physical appearance, internal makeup, etc • Building blocks = nucleotides • Nucleotide = sugar (5C), phosphate and a nitrogenous base • In DNA, deoxyribose, phosphate, and one of four N bases (adenine, thymine, cytosine, guanine)
IV. NUCLEIC ACIDS cont’d • RNA = ribose, phosphate group, and one of 4 N bases (adenine, uracil, cytosine and guanine) • DNA = double helix; RNA single stranded • Over 100 million base pairs in one human DNA molecule • Base pairing rules • In DNA bases joined together by H bonds to make up rungs of ladder