Organizing Elements
Organizing Elements. ** You need to add what is in RED to your notes. Mendeleev. He noticed that some elements had similar chemical and physical properties. He wrote each elements name and known information on cards and tried various arrangements.


Organizing Elements
E N D
Presentation Transcript
Organizing Elements ** You need to add what is in RED to your notes
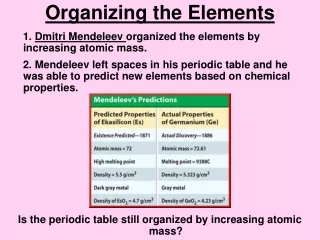
Mendeleev • He noticed that some elements had similar chemical and physical properties. • He wrote each elements name and known information on cards and tried various arrangements. • He realized that a pattern of properties appeared when he arranged the elements in order of increasing atomic mass. • He then predicted the properties of elements that would later be discovered.
The modern Periodic table • Periodic- In a regular, repeated pattern. • In 1913 Mosley discovered how to measure the number of protons in an atom. • Shortly after the table was rearranged in order of atomic number. • This caused some elements to shift position and the patterns of properties to become more regular.
Organization of Periodic Table • The properties of an element can be predicted from its location in the periodic table. • Periods- The rows in the periodic table. • As you move across a period, the elements’ properties change in a predictable way. • Ex: Reactivity, metals, nonmetals, gas. • Groups- The columns in the periodic table. • Also known as families. • The elements in each group have similar characteristics. • Ex: All elements in Group 1 react violently with water. The elements in Group 18 rarely react at all. The elements in the same group have the same number of valence electrons
Metals • Physical Properties- • Shininess • Malleability- material can be hammered or rolled into sheets. • Ex: • Ductility- Material can be pulled out into a long wire. • Ex: • Conductivity- The ability of a material to transfer heat or electricity. • Ex: • Others include: Color, Magnetism, and State.
Metals • Chemical Properties- • Reactivity- The ease and speed that an element combines, or reacts, with other elements. • Ex: • Sodium Reacting in water • Corrosion- the slow combination of a substance with oxygen. Iron rusting.
Alkali Metals • Group 1 • Key Properties: • Soft • React Violently • Low Melting Point • Low Density • Never found uncombined in nature • Have 1 valence electron and like to give it away
Alkaline Earth Metals • Group 2 • Key Properties: • Shiny grey-white • Relatively high melting and boiling points • Good conductors • Tarnish quickly when exposed to air • Never found uncombined in nature • Have 2 valence electrons that they need to give away
Transition Metals • Groups 3 through 12 • Key Properties: • Hard • Shiny • Good conductors of electricity. • Not very reactive
Lanthanides • Top row of elements grouped below the table. • Key properties: • Soft, Malleable, Shiny, High Conductivity. Actinides • Bottom row of elements grouped below the table. • Key properties: • Only Ac, Th, Pa, and U occur naturally. • The rest are synthetic.
Metals in Mixed groups • Some of the elements in Groups 13 through 15 are metals. • Ex: Aluminum, Tin. Synthetic Elements • Elements with an atomic number higher than 92. • They are not found naturally on earth. • They are made –or synthesized- when nuclear particles are forced to crash into one another. • Scientist use a Particle Accelerator to cause this.
Nonmetals • An element that lacks most of the properties of a metal. All of the yellow elements on the periodic table in class are Nonmetals. • Key Properties: Physical: • Poor conductors of electricity and heat. • 10 of the 16 nonmetals are gases. • Solid nonmetals are dull (not shiny) and brittle (not malleable or ductile). • Lower densities Chemical: • Most are reactive.
Carbon Family • Group 14 • A mixed group • Carbon- All organisms are made from carbon compounds. • Have 4 valence electrons. They need to add or remove 4. Nitrogen Family • Group 15 • A mixed group • The atmosphere is 80% Nitrogen. • Have 5 valence electrons. They need 3.
Oxygen Family • Group 16 • A mixed group • Oxygen and Sulfur are the most common elements in the family. • Have 6 valence electrons. They need 2. Halogen Family • Group 17 • The word Halogen means “salt forming” • 4 of the 5 are Nonmetals. Astatine is a Metalloid. • Most reactive Nonmetals • Have 7 valence electrons. They need 1.
Diatomic Molecules • Consist of two atoms. • The seven diatomic elements are: • Hydrogen, Nitrogen, Oxygen, Fluorine, Chlorine, Bromine, and Iodine. • 7TH Heaven Noble Gases • Group 18 • VERY stable. • They do not ordinarily form compounds. • Have a complete shell of valence electrons. They need 0.
Hydrogen • The simplest and smallest atom. • 1 proton, 1 electron. • Not grouped into any family because its properties are so different. • Make ups 90% of the atoms in the universe, but only 1% of the mass of the earth’s crust, oceans, and atmosphere. • Rarely found uncombined on earth. Most is combined with oxygen in H2O. • Most is located in stars and gas planets in the plasma state. • Has 1 valence electrons. It only needs 1 more to complete the first shell that can only hold 2 electrons. Most often shares.
Metalloids • Seven elements that are found along the “stairs” on the periodic table. The pink elements on the periodic table in class. • Have some characteristics of both metals and nonmetals. • Solid, brittle, hard, somewhat reactive • Are semiconductors • Substance that can conduct electricity under some conditions but not under other conditions. • Used to make computer chips, and lasers.