Organizing the Elements
Organizing the Elements. The Periodic Table. Elements. There are 118 different kinds of atoms. Most matter is made up of two or more kinds of atoms, but some consist of just one type of atom. When matter is composed of one type of atom it is called an element.

Organizing the Elements
E N D
Presentation Transcript
Organizing the Elements The Periodic Table
Elements • There are 118 different kinds of atoms. • Most matter is made up of two or more kinds of atoms, but some consist of just one type of atom. • When matter is composed of one type of atom it is called an element. • 94 elements are naturally occurring and the remaining are synthetic or unstable elements (24)
Elements Single atom of oxygen Molecule of oxygen
Element… • Element: An element is a pure substance that cannot be broken down into a simpler chemical substance by any physical or chemical means • Ex. Gold, copper, carbon, lead
Representing Elements… • Element Symbol: An abbreviation for a chemical element. • The symbols for elements are the same all over the world, regardless of the language. • The symbol of an element is taken from its name. Often the first letter is used. (Fluorine = F)
The name of an element may begin with the letter of a symbol already in use. So they are named by: • The element’s first two letters, Os, for Osmium • The first letter and some other letter in the name, Cl, for Chlorine • Symbols are taken from their Greek or Latin names , Aurium for gold • Symbols are sometimes named after famous scientists or places eg. Einsteinium or Americium
Rules for writing symbols: • The symbol always begins with a capital letter • If there is a second or third letter, it is always a lowercase letter • There is no period at the end of the symbol
Early Classification of Elements… • Alphabetical, colour, taste didn’t work • Metals vs. non-metals • These just didn’t seem to cut it….
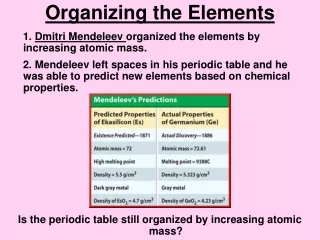
Mendeleev • Russian chemist • Organized 64 elements known at that time • He did it by atomic mass • Found similarities in physical properties of those in the same columns
Today’s Table • Periodic table: a table in which the elements are organized into rows and columns according to their atomic numbers and properties
Today’s Table • Primarily organized by; • atomic number, • metal/non-metal • properties like reactivity • Organized into groups/families and periods
The periodic Table of Elements… http://www.chemicool.com/ http://www.youtube.com/watch?v=GFIvXVMbII0
Metals • Metals: an element that is lustrous, malleable, ductile and conducts heat and electricity • Even if the metal appears to be dull in colour on the surface, freshly cut a metallic sheen can be seen • Located on the left side of the periodic table
Non-metals • Non-metals: an element usually a gas or dull powdery solid that does not conduct heat or electricity. • Upper right portion of the periodic table • Bromine is the only liquid non-metal at room temperature
Metalloids • Metalloids: an element that has properties of both a metal and a non-metal. Found along the staircase line between metals and non-metals
Periods (there are 7) • Each row represents a separate period • Have the same number of electron shells (energy levels)
Groups (there are 18) • Arranged together in rows (numbered 1 to 18) • Also called a family • All elements in each group have similar properties
Group 1: Alkali Metals • React rapidly when exposed to air and water • Shiny, silvery and soft • Highly reactive, often explosive in water http://www.youtube.com/watch?v=uixxJtJPVXk
Group 2: Alkaline Earth Metals • Be, Mg, Ca, Sr, Ba, Ra • Shiny and silvery, but not soft • Less reactive than alkali metals • These metals have a distinguishable flame color, Mg & Be white, brick-red for Ca, magenta-red for Sr, green for Ba and crimson red for Ra. http://www.youtube.com/watch?v=B2ZPrg9IVEo
Group 3-12:Transition Metal • Includes Ga, In, Sn, Tl, Pb, Bi, Lu, Lr and all the Uu… metals • Low reactivity • Rare earth metals/ inner transition metals often included
Group 18: Noble Gases • Unreactive, colourless, odourless • All gases (He, Ne, Ar, Kr, Xe, Rn) • He- low density for balloons, • Used for neon signs(glow with electric current) • Ar: blue, Kr: pink/white, Xe: purple, Ne:red http://www.youtube.com/watch?v=AmTZPx1whUQ
Group 17: Halogens • Most reactive non-metals • Rarely found in elemental form • Poisonous in large amounts • Many common uses: • Chlorine: pools • Iodine: clean wounds • Bromine: halogen light bulbs • http://www.youtube.com/watch?v=u2ogMUDBaf4
Hydrogen? • As you may have noticed hydrogen is not classified with any other group • Hydrogen is unique in the sense that it has a single electron in the first orbital • Hydrogen can donate an electron like the alkali metals but can accept an electron like a halogen • Non-metal
Periodic Trends • We saw changing reactivity between groups • Group 1 more reactive than group 2 • Group 17 more reactive than group 16 in the same row • Other trends:
Other trends • As you go down a family, the number of electron orbits increases • Within a family all elements have the same # of electrons in the outer most shell • Reactivity is based on the number of electrons in the outer shell