Organizing the Elements:
Organizing the Elements:. http://www.youtube.com/watch?v=pd8q9v5wCho. Objectives:. - Explain how Mendeleev discovered the pattern that led to the periodic table. -Identify the data about elements found in the periodic table. -Explain how the periodic table is useful. Dmitri Mendeleev .

Organizing the Elements:
E N D
Presentation Transcript
Organizing the Elements: http://www.youtube.com/watch?v=pd8q9v5wCho
Objectives: - Explain how Mendeleev discovered the pattern that led to the periodic table. -Identify the data about elements found in the periodic table. -Explain how the periodic table is useful
Dmitri Mendeleev He knew that some elements (substance that cannot be broken down into simpler substances by chemical means) had similar chemical and physical properties. He thought that these similarities were important clues to a hidden pattern.
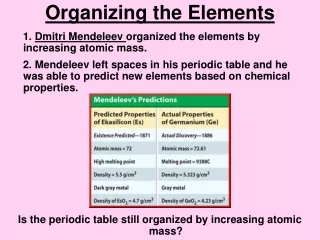
To find that pattern, Mendeleev wrote each element's melting point, density, and color on an individual card. He also included the element's atomic mass. Atomic Mass- number of protons and neutrons. Using this information, he tried arranging the cards in different ways. He noticed that a pattern of properties appeared when he arranged the elements in order of increasing atomic mass. He found that the properties repeated regularly. He then formed groups of elements that shared similar properties.
Mendeleev is credited for creating the first periodic table in 1869. Aperiodic table is an arrangement of elements showing the repeating pattern of the properties.
Before we can understand what the periodic table tells us, we must know something about atoms, too. An atom is the basic unit of a chemical element. In essence, everything is made up of atoms. Atoms are made up of even smaller parts...
At the very center of an atom, is the nucleus. Within the nucleus are protons and neutrons. Every atom of the same element has the same number of protons, this is called the atomic number. For example, the element Potassium has an atomic number of 19. This means that in every atom within Potassium, there are 19 protons in the nucleus.
The combined mass of protons and neutrons, make up the atomic mass of an element. Protons: Positive charge (+) Neutron: NO Charge (neutral)
Moving around in the space outside the nucleus are negatively charged particles called electrons.
What information does the Periodic Table contain, and how do I read it? 1. Atomic Number 2. Chemical Symbol 3. Atomic Mass
The periodic table has 7 periods, which form 18 columns/groups (families) The groups are numbered from Group 1 on the left to 18 on the right. The pattern of properties repeats in each period, so the elements in each group have similar characteristics. That's why they are called families!
How is the Periodic Table Useful? Notice that the atomic numbers increase from left to right. Also notice that each color-coded region corresponds to a different class of elements- metals, nonmetals, gases, etc. An element's properties can be predicted from its location in the periodic table, which is why chemists use it.
We will look specifically at: Metals Nonmetals Metalloids
Objectives: - Summarize the properties of metals -Describe how metals are classified on the periodic table Metals
What do you notice about the amount of metals in the periodic table? It's hard to imagine life without metals. Cars have iron (Fe), airplanes are covered in aluminum (Al), copper (Cu) wires carry electric current from lamps, stereos, and computers.
Elements are classified by their properties, including melting temperature, density, hardness, and thermal/electrical conductivity. Quick Info: Metals are elements that are good conductors of electricity, current, and heat. They also tend to be shiny and bendable - like copper wire for instance. The majority of elements in the periodic table are metals, and they begin on the left side extending across the periodic table.
Metal Physical Properties: 1. Luster 2. Malleability 3. Ductility 4. Conductivity Shiny and reflective Thermal Conductivity: The ability of an object to transfer heat. can be hammered or rolled into sheets or other shapes Can be pulled out or drawn into long wires (copper) * It does not take much thermal energy to raise the temperature of a metal. * Some metals are magnetic such as iron, cobalt, and nickel *Most metals are solid at room temperature * Mercury is the only metal that is liquid at room temperature Electrical Conductivity: The ability of an object to carry electric current.
Did you know???? Don't judge a coin by its coating! The U.S. penny, made of copper plated zinc, is just 2.5% copper by mass. The U.S. nickel is actually 75% copper, and the dime and quarter contain about 92% copper.
Metal Chemical Properties: Reactivity: The ease and speed with which an element combines, or reacts, with other substances. Metals react by losing their electrons to other atoms. Some metals are very reactive. For example, Sodium (Na) reacts strongly with water. However, gold (Au) and Platinum (Pt) do not react very easily. Still, some metals fall somewhere in between. Iron (Fe), reacts slowly with oxygen in the air forming rust. Corrosion: The deterioration of a metal due to a chemical reaction
Metals in a group (or family) have similar properties. Properties within a group change gradually as you look across the periodic table. For example, the reactivity of metals tends to decrease from left to right across the table. There are several classifications of metals in the periodic table: Alkali metals, alkaline earth metals, transition metals, metals in mixed groups, lanthanides, and actinides. You do not need to know these in depth, but please know that metals are separated into further classifications.
Objective: -Summarize the properties of nonmetals and describe the families that contain them.
Life on Earth depends on many nonmetals. It may seem weird to think about them in this way, but carbon (C), nitrogen (N), phosphorous (P), hydrogen (H), and oxygen (O) are all nonmetal elements in our bodies and on Earth. Some nonmetals are essential to life, others are poisonous and highly reactive, still others are nonreactive. Nonmetals are elements that lack the properties of a metal. With the exception of hydrogen, the non-metals are found on the right side of the periodic table. C H N P O
Nonmetal Physical Properties: 1. In general, most nonmetals are poor conductors of electric current and heat. 2. Solid nonmetals tend to be dull and brittle. 3. If you were to hit most solid nonmetals with a hammer, they would break or crumble into a powder. 4. Nonmetals usually have a lower density than metals. 5. Many nonmetals are gases at room temperature. 6. Some, like carbon, sulfur, and iodine, are solids at room temperature. 7. Bromine is the only nonmetal that is a liquid at room temperature.
Nonmetal Chemical Properties: Atoms of nonmetals usually gain or share electrons when they react with other atoms. Some nonmetals share their electrons... Water is an example of this. (H20)
Properties of Metals Properties of Nonmetals
Just like with the metals, there are also specific classifications of nonmetals. They are: carbon family, nitrogen family, oxygen family, halogen family, noble gases, and hydrogen. We will only specifically talk about the noble gases.
The noble gases are in group 18 on the periodic table. They are highly unreactive. Some examples are Neon and Helium. They are "noble" because that is how royalty was viewed... aloof and non-interactive with other people.
Between the metals and the nonmetals in the periodic table are the metalloids. They are unique, and have some properties of metals and some properties of nonmetals... -All metalloids are solids at room temperature - They are brittle, hard, and somewhat reactive - The most common is Silicon -Their most useful property is the ability to conduct electric current. Their conductivity depends on temperature and exposure to light. For this reason, metalloids like silicon and germanium are used to make semiconductors. Semiconductors are substances that can conduct electric current under some conditions, but not under others.
Coloring the Periodic Table Handout pg. 16 of The Periodic Table Printable Periodic Table PDF Families of ELements on the Periodic Table from TpT Periodic table scavenger hunt quiz Extra credit - meet the elements packet