Organizing the Elements 6.1
Section 6.1-1. Organizing the Elements 6.1. History of the Periodic Table. Organizing the Elements. 6.1-1.

Organizing the Elements 6.1
E N D
Presentation Transcript
Section 6.1-1 Organizing the Elements 6.1 History of the Periodic Table
Organizing the Elements 6.1-1 • In a self-service store, the products are grouped according to similar characteristics. With a logical classification system, finding and comparing products is easy. You will learn how elements are arranged in the periodic table and what that arrangement reveals about the elements.
6.1-1 Searching For an Organizing Principle • Searching For an Organizing Principle • How did chemists begin to organize the known elements?
6.1-1 Searching For an Organizing Principle • Chemists used the properties of elements to sort them into groups.
6.1-1 Searching For an Organizing Principle • Chlorine, bromine, and iodine have very similar chemical properties.
6.1-1 History of the Periodic Table • Antione-Laurent de Lavoisier (1789) - Produced the first modern chemical textbook. It contained a list of elements, or substances that could not be broken down further. It also forms the basis for the modern list of elements. His list, however, also included light and caloric, which he believed to be material substances. However, as Lavoisier's descriptions only classified elements as metals and non-metals, it fell short of a complete analysis.
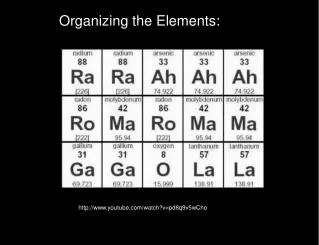
6.1-1 History of the Periodic Table • Johann Wolfgang Döbereiner (1817) – Johann began to formulate one of the earliest attempts to classify the elements. He found that some elements formed groups of three with related properties. He termed these groups “triads“. Some triads classified by Döbereiner are: chlorine, bromine, and iodine; calcium, strontium, and barium; sulfur, selenium, and tellurium; and lithium, sodium, and potassium. • In all of the triads, the atomic weight of • the second element was almost exactly • the average of the atomic weights of the • first and third element.
6.1-1 History of the Periodic Table • Alexandre-Emile Béguyer de Chancourtois (1862) - was the first person to notice the periodicity of the elements — similar elements seem to occur at regular intervals when they are ordered by their atomic weights. • He devised an early form of periodic table, which he called the telluric helix, with the elements arranged in a spiral on a cylinder by order of increasing atomic weight.
6.1-1 History of the Periodic Table • In this method, de Chancourtois saw that elements with similar properties lined up vertically. His paper was published in 1862, but used geological rather than chemical terms and did not include a diagram; as a result, it received little attention.
6.1-1 History of the Periodic Table • John Newlands (1865) - Classified the fifty-six (56) elements that had been discovered at the time into eleven groups which were based on similar physical properties. • Newlands noted that many pairs of similar elements existed which differed by some multiple of eight in • atomic weight. However, his law of octaves, likening this periodicity of eights to the musical scale, was ridiculed by his contemporaries because it was not scientific. It was not until the following century, that the importance of the periodicity of eight would be accepted.
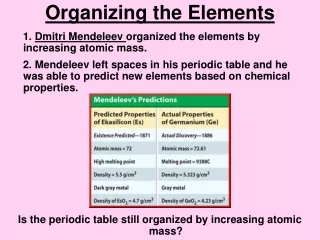
6.1-1 History of the Atom • DimitriMendeleev (1869) - was the first scientist to make a periodic table much like the one we use today. Mendeleev arranged the elements in a table ordered by atomic weight. • On March 6, 1869, a formal presentation was made to the Russian Chemical Society. His table was published in an obscure Russian journal but quickly republished in a well-known German journal, • ZeitschriftfürChemie (Eng., "Chemistry Magazine"), in 1869.
6.1-1 History of the Atom • Mendeleev’s Periodic Table
6.1-1 History of the Atom • At about the same time, a German Chemist, LotharMeyer was also working on a periodic table. Although his work was published in 1864, and was done independently of Mendeleev, few historians regard him as an equal co-creator of the periodic table. • For one thing, Meyer's table only included 28 elements. Furthermore, Meyer classified elements not by atomic weight, but by valence alone. • Finally, Meyer never came to the idea of predicting new elements and correcting atomic weights like Mendeleev did.
6.1-1 History of the Atom • However, most scientists agree that , by itself, Mendeleev's accurate prediction of the qualities of the undiscovered elements lands him the larger share of credit. In any case, at the time Mendeleev's predictions greatly exceeded those of Lothar Meyer. • Lothar Meyer
6.1-1 History of the Atom • Henry Moseley (1914) - found a relationship between an element's X-ray wavelength and its atomic number, and therefore re-sequenced the periodic table by nuclear charge (atomic number) rather than atomic weight. • Before this discovery, atomic numbers were just sequential numbers based on an element's atomic weight. Moseley's discovery showed that atomic numbers had an experimentally measurable basis.
6.1-1 Mendeleev’s Periodic Table • Mendeleev’s Periodic Table • How did Mendeleev organize his periodic table?
6.1-1 Mendeleev’s Periodic Table • Mendeleev arranged the elements in his periodic table in order of increasing atomic mass. • The periodic table can be used to predict the properties of undiscovered elements.
6.1-1 Mendeleev’s Periodic Table • An Early Version of Mendeleev’s Periodic Table