1 / 6
Télécharger la présentation 

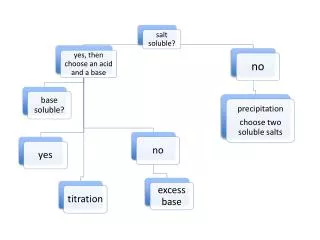
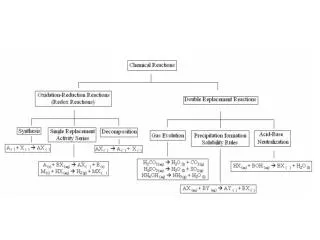
Solubility Rules
An Image/Link below is provided (as is) to download presentation
Download Policy: Content on the Website is provided to you AS IS for your information and personal use and may not be sold / licensed / shared on other websites without getting consent from its author.
Content is provided to you AS IS for your information and personal use only.
Download presentation by click this link.
While downloading, if for some reason you are not able to download a presentation, the publisher may have deleted the file from their server.
During download, if you can't get a presentation, the file might be deleted by the publisher.
E N D
Presentation Transcript
Solubility Rules 10th Grade ATC: Tuesday January 24th
Solubility Rules • Not all solutes dissolve • Use General Solubility Rules to determine solubility. • Compare the problem to the solubility rules chart
Practice Questions • Ca(NO3)2 Soluble (salts containing NO3- are soluble) b) FeCl2 Soluble (all chlorides are soluble) c) Ni(OH)2 Insoluble (most hydroxides are insoluble)
Assignment • Turn in the following 2 Questions to your science teacher
Question 1 • Which of the following salts has the greatest solubility in water at 25°C? • F CaCO3 • G FeS • H HgCl2 • J KClO4
Question 2 • Which of the following salts has the least solubility in water at 25°C? FCaCO3 G KBr H CaCl J KI
More Related