Refinery Basics
Refinery Basics. John Kulluk Ph.D. Torrance Fire Department & Robert Distaso PE – 714/573-6253 Orange County Fire Authority. Refining Means. . . 1. To reduce to a pure state, to remove impurities 2. To improve or perfect. Refinery Process Flow Chart. Sulfur. Gas Plant. Fuel Gas. Isom.


Refinery Basics
E N D
Presentation Transcript
Refinery Basics John Kulluk Ph.D. Torrance Fire Department & Robert Distaso PE – 714/573-6253 Orange County Fire Authority
Refining Means. . . 1. To reduce to a pure state, to remove impurities 2. To improve or perfect
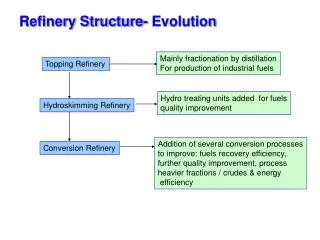
Refinery Process Flow Chart Sulfur Gas Plant Fuel Gas Isom Distilling Splitter Gasoline Reformer Jet Fuel Hydrotreating Distillate Fuel Hydrotreating Alky CCU Flasher ResidualFuel Visbreaker Leffler, 1985
Other Refinery Units • Steam Generation • Wastewater Treatment • Hydrogen Generation • Power Generation (e.g., cogen) • Air Separation Plant • Loading/Unloading - Railcar, Trucks, etc. • Storage (high pressure hydrocarbon, crude oil, intermediates) • Floating-Roof Tanks - 150‘ diameter is common • Spherical Tanks - 50‘ are common • Horton Spheroid (refrigerated) • Steam-Heated Tanks for “Heavier” Products • Self-Contained Firewater Supply • Firewater Pumps
What is Crude Oil? • Mixture of organic carbon chain molecules • Impurities include sulfur and nitrogen compounds • Some metals and salts too
Straight-Chain Hydrocarbons Olefins Cyclic H/C Aromatics (Benzene, toluene, xylenes) Mercaptans Hydrogen Sulfide (H2S) Greases Propane LPG Components such as . . .
Other Hazardous Materials • Sulfur • Hydrofluoric Acid (HF) • Sulfuric Acid (H2SO4) • Ammonia (NH3) • Sodium Hypochlorite • Radioactive Materials • Chlorine • Amines • MEK • Sulfur Dioxide (SO2) • Heavy Metal Catalysts • Sour Water • Caustic (fresh/spent) • Alcohol • Asbestos
What Goes on at a Refinery. . .? • Separation of components by distillation, e.g.: • Atmospheric • Vacuum • Hydrotreating (uses excess hydrogen) • Breaking apart molecules to make smaller ones, e.g.: • catalytic cracking • hydrocracking • Joining molecules to make bigger ones, e.g.: • Reforming - alkylation that lengthens the hydrocarbon chain • Reforming - cyclic that generates hydrogen
Physical Hazards • High Pressure/Temperature Steam • Oil/Gas-Fired Furnaces • Acoustic • High Voltage (4160V, 480V, 13.2 kV) • Falling Hazards • Confined Space Hazards • Cranes/Lifting Hazards • Hot Work Hazards • Acid Exposure • Toxic Vapors • Radiation • Flammability Hazards
Common PPE Requirements • Hardhat • Hardsoled / Hardtoe Shoes • Safety Glasses with Side Protection • Safety Goggles or Faceshield • Fire-Resistant Clothing
Process Hazards • Emergency Flare • Atmospheric Pressure Relief • High Temperature (up to 2000oF) • Low Temperature (e.g., Brittle Fracture) • High Pressure (up to 3000 psig) • Low Pressure (e.g., vacuum)