Enzymes
Enzymes. Topic 3.6 and AHL 7.6. http://www.livingscience.co.uk/joomla/index.php?option=com_content&view=article&id=57:enzymes&catid=42:enzymes&Itemid=54. I.A. Investigation: Factors affecting enzyme activity investigations.


Enzymes
E N D
Presentation Transcript
Enzymes Topic 3.6 and AHL 7.6 http://www.livingscience.co.uk/joomla/index.php?option=com_content&view=article&id=57:enzymes&catid=42:enzymes&Itemid=54
I.A. Investigation: Factors affecting enzyme activity investigations You will be looking at the decomposition of H2O2 by catalase in yeast. How could you get catalase? What is catalase? Timeline: Two periods to investigate different lab protocols. Your ORIGINAL lab design will be due the following day. Class period to work on lab design with students using a similar design (I’ll explain). Two lab periods for YOUR INDIVIDUAL data collection. Due one week later
This is how you will be graded. Remember to use the report format you have used before. I’ll put a link here to it in the future on the website I haven’t set up.
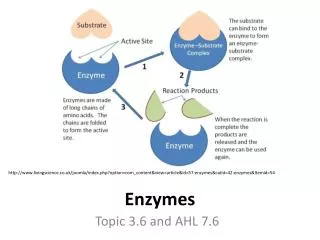
Enzymes, substrates and active sites Substrate: reactant in a biochemical reaction. Enzyme: globular protein which acts as a catalyst for biochemical reactions. Polar regions of amino acids attract substrate & active site of the enzyme. Active site: region on the surface of an enzyme to which substrates bind and which catalyzes the reactions. Once a substrate has been locked into the active site, the reaction is catalyzed. The products are released and the enzyme is used again. It is NOT consumed in the reaction. http://i-biology.net/
http://www.northland.cc.mn.us/biology/biology1111/animations/enzyme.swfhttp://www.northland.cc.mn.us/biology/biology1111/animations/enzyme.swf http://highered.mcgraw-hill.com/sites/0072495855/student_view0/chapter2/
Enzymes are specific to their substrate! The Lock-and Key hypothesis: the substrate and the active site match each other in two ways: Structurally – the 3D structure of the active site will only fit the substrate. No fit, no Rxn. Chemically – substrates that are not chemically attracted to the active site won’t be able to react. http://www.youtube.com/watch?v=AKyyvuOmXq0 http://i-biology.net/
The induced –fit model better explains enzyme activity If the lock-and-key model were true, one enzyme would only catalyze one reaction. In reality, some enzymes can catalyze multiple reactions. As the substrate approaches the enzyme, it induces a conformational change in the active site- it changes shape to fit the substrate. This stresses the substrate, reducing the activation energy of the reaction. Conformational change Enzyme reverts to original shape http://www.stolaf.edu/people/giannini/flashanimat/enzymes/enzyme.swf
Enzymes lower the activation energy of a reaction!! http://www.biology.arizona.edu/biochemistry/problem_sets/energy_enzymes_catalysis/01t.html Activation energy is the amount of energy that must be put into a reaction to make it occur. Click on the graph to link to the Univ. of Arizona site.
http://www.stolaf.edu/people/giannini/flashanimat/enzymes/transition%20state.swfhttp://www.stolaf.edu/people/giannini/flashanimat/enzymes/transition%20state.swf An enzyme stresses the bonds in the substrates, reducing the activation energy required for a reaction to occur.
Denaturation Enzymes are globular proteins. Their structure can be altered by changes in pH or temperature – if the shape of the active site is changed substantially, they will not function. http://highered.mcgraw-hill.com/sites/0072943696/student_view0/chapter2/animation__protein_denaturation.html
Denaturation • High temperatures cause denaturation as the extra energy leads to increased vibration, breaking intra-molecular bonds. • Changes in pH cause denaturation as hydrogen bonds are broken. • Both methods result in an altered 3D structure of the active site, and this change is irreversible. http://www.youtube.com/watch?v=3IL_Df5ouUc
Factors affecting enzyme activity: Use this animation to test the affect of the following factors on enzyme activity: Temperature pH Substrate concentration When you have finished this, complete the enzyme activity sheet. http://www.kscience.co.uk/animations/model.swf
The Effect of Temperature on Enzyme Activity As temperature increases, rate of reaction increases as molecules have more energy, move faster and therefore collide and react more frequently. Above optimal temperature any more increase denatures the protein. The active site changes, and the enzyme is non-functional. A thermophile, such as bacteria in boiling pools (think Yellowstone National Park) can withstand MUCH higher temps before it denatures.
The Effect of Temperature on Enzyme Activity Try this virtual lab:
The Effect of Temperature on Enzyme Activity Enzymes only operate within a narrow pH range. This is called the optimum pH. Look at this example of two digestive enzymes. If there is a deviation from the optimum pH, the hydrogen bonds between the amino acids in the structure of the enzyme are broken. The result is the loss of shape of the active site of the enzyme, so it does not function. This is usually a permanent change.
http://users.rcn.com/jkimball.ma.ultranet/BiologyPages/D/DenaturingProtein.htmlhttp://users.rcn.com/jkimball.ma.ultranet/BiologyPages/D/DenaturingProtein.html
The effect of substrate concentration on enzyme activity At the optimum concentration of substrate molecules, all active sites are full and working at maximum efficiency. Increasing substrate concentration increases the rate of reaction Any increase in concentration beyond the optimum will nave no added effect as there are no extra active sites to be used.
Lactose intolerance http://i-biology.net/ Lactose (milk sugar) can cause allergies in some people. This is usually because they can’t produce the enzyme lactase in quantity. No ice cream for YOU! http://www.cartoonstock.com/directory/l/lactose.asp
Most people produce less lactase as they get older. Think about it…we don’t live off milk after we are weaned from mom. In some regions, such as Europe, a mutation has allowed lactase production to continue into adulthood. Those who are lactose intolerant don’t have this mutation.
How do those who are lactose intolerance cope? Take a lactase supplement – these are produced from a fungus called Aspergillusniger Drink lactose free milk – milk treated with lactase (produced by A. niger) • Lactose-free milk is made by different methods: • Add lactase to milk • Run milk though apparatus with immobilize lactase. Uses alginate beads, no enzyme in final product. A. niger
Try this: by changing just one letter at a time, get from ‘Tread’ to ‘Blink’. All intermediates must be real English words.
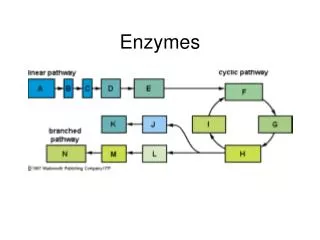
Metabolic pathways* are chains of enzyme catalyzed reactions. The product of one reaction is a reactant in the next. *or biochemical pathways Initial substrate Intermediates End product http://highered.mcgraw-hill.com/sites/0072943696/student_view0/chapter2/animation__a_biochemical_pathway.html
The Krebs Cycle (cell respiration) and Calvin Cycle (photosynthesis) are examples of enzyme catalyzed, metabolic pathways. http://www.sparknotes.com/health/carbohydrates/section3.rhtml http://library.thinkquest.org/C004535/calvin_cycle.html
Enzymes can be inhibited by other molecules Inhibition can be competitive or non-competitive Won’t Fit! Blocked! Inhibitor fits into the allosteric site* causing a conformational change in the active site. The substrate cannot attach to react. Inhibitor fits the active site and prevents the substrate from entering. *allosteric site = Other site
Competitive Inhibition A competitive inhibitor blocks the active site, preventing the substrate from entering. The higher the concentration of inhibitor, the slower the rate of reaction Even with competitive inhibition, the same maximum rate of reaction will be achieved if more substrate is added because we have not change the number of enzymes available.
Overcoming alcoholism: An example of competitive inhibition Normal metabolism of ethanol (alcohol): Antabuse (disulfiram) competes with the aldehyde oxidase and prevents the acetaldehyde from being converted to acetic acid. A build up a acetaldehyde follows, resulting in a strong feeling of nausea and other hang over symptoms. A great deterrent from drinking!! Antabuse is administered as a daily pill, so how well it works depends on how much a person is motivated! If they stop taking it they can drink again.
Non-competitive Inhibition – inhibitors bind to an allosteric (other) site on the enzyme. This binding alters the active enzyme site and substrates can no longer bind. As the concentration of inhibitor increases, the rate of reaction decreases, because there are fewer active sites available for the reaction.
Let’s take a look at how non-competitive inhibition would look. http://www.stolaf.edu/people/giannini/flashanimat/enzymes/allosteric.swf
Let’s take a look at how inhibition disturbs the final product of biochemical pathways. http://highered.mcgraw-hill.com/olcweb/cgi/pluginpop.cgi?it=swf::535::535::/sites/dl/free/0072437316/120070/bio10.swf::