Radioactivity
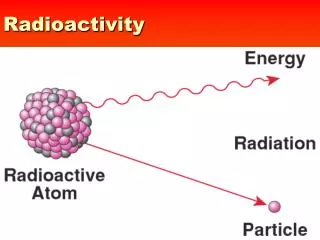
Radioactivity. Radioactivity is the spontaneous decay of unstable nuclei to release either alpha or beta particles or gamma rays and energy.

Radioactivity
E N D
Presentation Transcript
Radioactivity is the spontaneous decay of unstable nuclei to release either alpha or beta particles or gamma rays and energy
Many elements (red) exist primarily as radioactive elements, while others have both radioactiveand stable isotopes. After U, all are radioactive and manmade.
Why are some isotopes unstable (radioactive), while others are not? Larger elements have protons & neutrons too far apart to be held together by the strong nuclear force—acts at small distances. (Remember, p+ are repelled by each other)
Neutrons are held together by strong nuclear force, and they shield p+ from each other. So, stable isotopes of atoms with large protons numbers usually have more neutrons than protons—but if the nucleus gets too high in diameter, the strong nuclear force is too weak to hold it together. e.g., hydrogen-1 is stable, but Hydrogen-2 deuterium and Hydrogen-3 tritium are radioactive
The ratio of p+/no determines whether atoms of an isotope will be stable or radioactive. Green elements are stable, but yellow ones are radioactive.
Three main types of radioactive decay exist: Alpha α, Betaβ, Gamma Ɣ 224Ra --> 220Rn + 4 He2+ 2 alpha α particles are He nuclei (2 protons and 2 neutrons, mass 4; they have a positive 2 charge because they have no electrons 4 He 2+ 2 The large size of alpha particles makes them sluggish and slow and low in energy. You can protect yourself from α radiation by holding up a sheet of paper.
Beta decay: Gain or loss of a proton through conversion of neutrons to a proton or protons to a neutron—whichev er confers the stable ration; during the conversion, β particles about the size of an electron are released. β particles low masses allow them to travel at higher speed and to have higher energy—to protect yourself from these, you must use a plastic sheet ¼ inch thick. Gamma Ɣ decay—high energy waves, no particles; the highest energy of radiation—to protect yourself, you must use a 1/8 inch lead shield—like in the heavy apron at the dentist’s office from when you have Xrays.
Writing balanced chemical equations to represent nuclear decay.Reactants products Equations obey Law of conservation of matter & energy: Total mass of reactants = total mass products Total charge of reactants = total charge products Total charge of reactants = total charge products For α & β nuclear decay ONLY: Total #s and identities of elements on reactant side = totals & identities for product side. Why? Transmutation—creation of new elements when neutrons are lost are gained
Why can you know whether α or β decay occurred in each? Look at mass numbers & atomic numbers! thorium-232 --> radium-228 + α reduced mass radium-228 --> actinium-228 + β mass same, ↑atomic # actinium-228 --> thorium-228 + βmass same, ↑atomic # thorium-228 --> radium-224 + α reduced mass radium-224 --> radon-220 + α ? radon-220 --> polonium-216 + α ? polonium-216 --> lead-212 + α ? lead-212 --> bismuth-212 + β ? bismuth-212 --> polonium-212 + β ? polonium-212 --> lead-208 + α ?
Radioactive isotopes emit radiation that destroys or causes mutation of DNA—think of examples of how people are protected.
Nuclear energy takes advantage of the huge amount of heat energy released during radioactive decay Pros Cheap Lots of U-238 available Cons Long half life of wastes—no where to store them Accident possibility Cost of building and maintaining, even after closing Weapon grade plutonium Terrorist obtaining waste Heat water, release steam
Energy from decay heats water to turn turbines that generate electricity. Pellet (1 penny)=2 tons coal equivalent!
Biggest problem is storage of wastes with long half lives: time for half of a sample to become nonradioactive. After 10 half lives, safe disposal is possible.
If an isotope has a half life of 37,000 years, then how much of a 100 kg sample of it will still beradioactive after: 37000 years 74000 years 111000 years Ten half lives allows disposal
Plutonium needs storage for 400,000 years! Why so hard to store it? Heat decays containers & breaks rocks, allowing the waste to reach ground water and thus the food chain! High cost of protection from theft No long term site yet! Highest risk waste stored on site at the plants in swimming pools! (theft waiting to happen or spills)
Why possible accidents at nuclear reactors? Fission chain reactions get out of control. Normally, collisions with heavy water slow the reactions.
Retired power plants (30 years old or so) require as large a staff and as much protection as a working plant. Yet they generate no money or electricity! There’s not yet a place safe enough to store the used fuel rods. Now they’re stored under water at the plants! (also, transport is very dangerous)
Nuclear bombs use chain reactions to create huge shock waves: heat compresses air together to create blast & radiation damages DNA and RNA.
Why do we have greater risk from dirty bombs than from traditional atom bombs? Easier to build Any radioactive materials will work—long half lives work best Can detonate with explosives by blowing a plane up in the air and contaminate huge area Cost of clean up is massive—maybe not possible Move out of area to avoid long term exposure that can cause cancer and birth defects Unguarded waste and submarine fuel rods are scattered in large amounts in the former USSR. It’s hard to detect wastes—most ports, airports, borders don’t screen with geiger counters!
Fusion energy is the ultimate: fuse atoms together at high temperatures Would generate energy in enormous amounts starting with water as fast moving positrons released increase motion of water and increase its temperature steam to run electrical turbines. No wastes made Free! Problem: all materials melt at lower temperature—hard to reach the temperature to start it—magnetic “bottles” are the only container to hold the reaction mix!