Radioactivity
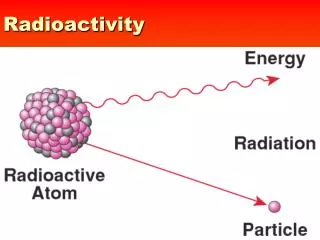
Radioactivity. A. The spontaneous breakdown of an unstable atomic nucleus with the release of radiation. III. Radioactivity. Definition: Unstable isotopes change into different, more stable elements.

Radioactivity
E N D
Presentation Transcript
Radioactivity A. The spontaneous breakdown of an unstable atomic nucleus with the release of radiation
III. Radioactivity Definition: Unstable isotopes change into different, more stable elements. Can change to different element, just mass, or both. When change occurs nuclear particles are released/absorbed What particles are in the nucleus?
Comparison Chemical Reactions Nuclear Reactions Atoms change to another element Protons, neutrons and other particles involved in changes Large changes in energy Measureable change in mass • Identity of atoms do not change • Electrons involved as bonds break & form • Small changes in energy • No measureable change in mass
Types of Radioactive Particles • Positron – same mass as electron, opposite charge (+1)
III. Fission • Definition: splitting a nucleus into two or more smaller nuclei • 1 g of 235U = 3 tons of coal
IV. Fusion • Definition: combining two nuclei to form one nucleus of larger mass • 40,000,000 K to sustain • Occurs naturally in stars • 1 g of fusion fuel = 20 tons of coal
Interesting Website • http://library.thinkquest.org/17940/texts/atom/atom.html
235U is limited danger of meltdown toxic waste thermal pollution fuel is abundant no danger of meltdown no toxic waste not yet sustainable Fission vs. Fusion FUSION FISSION
V. Half Life • Definition: Time required for half the atoms of a radioactive nuclide to decay. Shorter half-life = less stable
V. Half Life C. Examples: • If you start with 400 atoms of a radioactive substance, how many would remain after one half life? • … after two half lives? • … after three half lives? • … after four half lives? • How many half lives occur for a substance to decay 1/16th of its original mass?
Half Life • Gold-198 has a half life of 3 days. How much of 96gram sample will be left after 9 days? • If 100 grams of carbon-14 decays until only 25grams of carbon is left after 11,460 years, what is the half life of carbon-14?
5. Potassium-42 has a half life of 12.4 hours. How much of an 848g sample will be left after 62 hours?
How long is the half life for carbon-14? If only 25% of the carbon-14 remains, how old is the material containing carbon-14? If a sample originally had 120 atoms of carbon-14, how many atoms will remain after 16,110 years? If a sample known to be about 10,740 years old has 400 carbon-14 atoms, how many atoms were in the sample when the organisms died?