Radioactivity
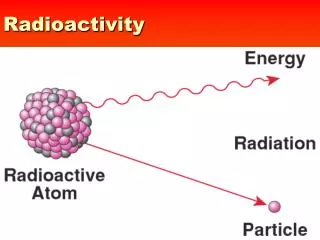
Radioactivity. Chapter 6.1. Radioactivity. Comes from an unstable nucleus Can be either… Particles or Energy (as electromagnetic radiation) or Both. Types of Radiation. Alpha Particle ( ) Equivalent to a helium nucleus Beta Particle () an electron Gamma Ray ()

Radioactivity
E N D
Presentation Transcript
Radioactivity Chapter 6.1
Radioactivity • Comes from an unstable nucleus • Can be either… • Particles • or Energy • (as electromagnetic radiation) • or Both
Types of Radiation • Alpha Particle () • Equivalent to a helium nucleus • Beta Particle () • an electron • Gamma Ray () • Electromagnetic radiation (like light, radio, etc) • Neutron Emission • a neutron
Nuclear Decay When an unstable nucleus emits an alpha or beta particle… …the number of protons or neutrons changes. This means that the identity of the atom may change also.
Alpha Decay • Happens when a nucleus gives up two protons and two neutrons. • Board Work
Beta Decay • Happens when a neutron splits and forms a proton and an electron. • The electron leaves the nucleus as radiation. • Board Work
Gamma rays are energy that may leave when other decay takes place. There is NO change in the atom from this radiation Neutron Emissions are neutrons that may leave when other decay takes place. There is NO change in the identity. There IS a change in mass of the atom. Other Radiation
Practice • Page 191 • Numbers 1 – 4 • Go!!!
Radioactive Decay Rates • Decay is spontaneous and random… • when observing ONE atom. • When observing a large sample of material the decay is no longer random.
Half-life • This is the time required for half of a sample to decay. • Knowing this info can be useful. • Examples • Uranium-238 4.47 x 109 years • Potassium-40 1.28 x 109 years • Carbon-14 5730 years • Iodine-131 8.1 days • Hafnium-156 0.025 seconds