Discovering Radioactivity: Key Moments & Insights
Explore the fascinating discoveries of radioactivity by luminaries like Roentgen, Becquerel, and the Curies. Understand types of nuclear radiation, band of stability, transmutation, and basic assessment questions in this informative chapter on nuclear chemistry. Witness the transformative power of radioactive decay and gain insights into the science behind it.

Discovering Radioactivity: Key Moments & Insights
E N D
Presentation Transcript
Radioactivity Chapter 25
Nuclear Radiation • Nuclear chemistry • study of the structure of atomic nuclei • changes they undergo.
The Discovery of Radioactivity • Wilhelm Roentgen (1845–1923) • 1895-invisible rays were emitted when electrons bombarded the surface of certain materials. • caused photographic plates to darken. • named the invisible high-energy emissions X rays.
The Discovery of Radioactivity • Henri Becquerel (1852–1908) was studying phosphorescence • minerals that emit light after being exposed to sunlight • phosphorescent uranium salts produced spontaneous emissions that darkened photographic plates.
The Discovery of Radioactivity • Marie Curie (1867–1934) and her husband Pierre (1859–1906) took Becquerel’s mineral sample (called pitchblende) and isolated the components emitting the rays. • darkening of the photographic plates was due to rays emitted specifically from the uranium atoms present in the mineral sample.
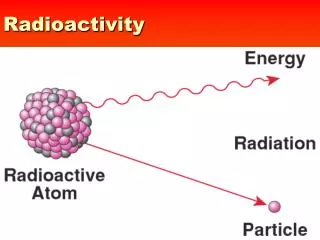
The Discovery of Radioactivity • Marie Curie named the process by which materials give off such rays radioactivity • the rays and particles emitted by a radioactive source are called radiation.
Types of Radiation • isotopes are atoms of the same element that have different numbers of neutrons. • Isotopes of atoms with unstable nuclei are called radioisotopes • emit radiation to attain more stable atomic configurations in a process called radioactive decay • lose energy by emitting one of several types of radiation.
Why do some atoms decay? • The nucleus contains tightly packed protons and neutrons (nucleons) • The strong nuclear force keeps the nucleons packed together even though protons want to push each other away • Stable atoms have a neutron to proton ratio of about 1:1
As atomic number increases, more neutrons are required to have enough of a strong force to keep the protons pushed together • The neutron to proton ratio for stable atoms increases to 1.5:1
Band of Stability • When the number of protons and neutrons are plotted, the stable nuclei are found within the “band of stability” • Radioactive isotopes are outside the band of stability • They will undergo nuclear reactions to become more stable • All elements higher than atomic# 83 are radioactive
Basic Assessment Questions Calculate the neutron-to-proton ratio for . Topic 26 Question 3
Types of Nuclear Radiation • Alpha • Beta • Gamma
Alpha Radiation • Release of 2 protons and 2 neutrons • Equivalent to a He nucleus • Charge of 2+ • Mass = 4 amu • Largest and slowest • Least penetrating can be stopped by paper • Changes to a different element with a lower atomic mass and lower atomic number • Example: Polonium-212 (atomic# 84) is converted to Lead-208 (atomic# 82)
Beta Radiation • Decay of a neutron into a proton and electron • Electron is emitted, proton stays • Forms a new element b/c of addition of proton • Decay of the proton into a neutron and positron (like a positive electron) • The positron is emitted as a beta particle • Faster than alpha particles can be stopped by aluminum foil
Gamma Radiation • Not a particle • Electromagnetic wave with short wavelength and high frequency & energy • No mass, no charge • Very fast speed of light • Stronger than X-ray • Stopped by several centimeters of lead
Transmutation: changing one element into another through radioactive decay • Adding or removing a proton changes the atomic number, resulting in a different element • Half-Life: amount of time for half of a sample of a radioactive element to decay into something else • Can range from a fraction of a second to billions of years • Amount remaining=initial amount(1/2)t/T • t=total time • T=half-life
Half-life mf:final mass mi:initial mass n:# of half-lives
Half-life • Fluorine-21 has a half-life of 5.0 seconds. If you start with 25 g of fluorine-21, how many grams would remain after 60.0 s?
parent nuclide alpha particle daughter nuclide Nuclear Decay • Alpha Emission Numbers must balance!!
electron positron Nuclear Decay • Beta Emission • Positron Emission
electron Nuclear Decay • Electron Capture • Gamma Emission • Usually follows other types of decay.
Types of Transmutation • Induced transmutation • Nucleus of an unstable isotope (radionuclide) is struck with a high velocity charged particle • Particle accelerator • Need lots of energy and unstable nucleus • Elements atomic 93 and higher (transuranium elements) • Natural transmutation • Occurs naturally as a radioisotope decays to become more stable
Basic Assessment Questions What element is formed when polonium-214 ( ) radioisotope undergoes alpha decay? Give the atomic number and mass number of the element. Topic 26 Question 1
Basic Assessment Questions What element is formed when undergoes beta decay? Give the atomic number and mass number of the element. Topic 26 Question 2
Basic Assessment Questions Topic 26 Question 4 Write a balanced nuclear equation for the beta decay of the following radioisotope.
Nuclear Fission • Fission = divide • Neutron hits an unstable atom • Nucleus splits into two fragments of about the same mass • Some single neutrons are released (energy) • These neutrons can smash into other atoms • Causes a chain reaction
Nuclear reactors • Nuclear power plants use the process of nuclear fission to produce heat in nuclear reactors. • The heat is used to generate steam, which is then used to drive turbines that produce electricity.
Little Boy: $2billion in research; made of Uranium-235; equal to 20,000 tons of TNT; 140,000 people died; 2/3 of the city destroyed • Fat Man: Plutonium-239; 70,000 people died; 40% of the city destroyed
Hydrogen Bomb • 1000 times more powerful than atomic bomb • March 1, 1954; Bikini Atoll in Pacific • Never in war • Fission reaction triggers fusion of Hydrogen isotopes
Nuclear Fusion • Opposite of fission • Two nuclei fuse together to form one nucleus with a larger mass • Not simple sum of masses • Some mass lost as energy • Requires high temperature: Thermonuclear reaction • Occurs in the sun and stars • 4 H combine to form one He, 2e- and energy
Radiation Detectors • Cloud Chamber • supersaturated water or ethanol • radioactive particle flows through and knocks e- off • vapor condenses showing path • alpha: short/thick trails; beta: long/thin • Bubble Chamber • superheated liquid • e- knocked off again • bubbles are formed
Measuring Radiation • Geiger Counter • produces electric current when near radiation • Results in clicks or a digital reading
Using Nuclear Reactions in Medicine • Tracers: monitor body processes • Iodine-131 • emits beta particles • used to detect tumors in thyroid gland • also used: Carbon-11 and Sodium -24
Cancer Treatment • damage cancer cells • Gold -198 or Iridium -192 -- implanted in or near tumor • Cobalt-60 • outside body • emits gamma rays
Positron Emission Tomography (PET) • Fluorine-18 attached to molecules that go to brain • positrons are emitted and collide with electrons forming 2 gamma rays • the gamma rays are detected and indicate brain activity
http://www.hpwt.de/Kern2e.htm • http://www.colorado.edu/physics/2000/isotopes/radioactive_decay3.html • http://www.msd.k12.or.us/schools/mhs/projects/Fission/frames.html • http://www.cnn.com/SPECIALS/cold.war/experience/the.bomb/history.science/