Radioactivity
Radioactivity. First it’s here, then it’s gone!. Occur when bonds are broken and formed. Atoms remain unchanged, though they may be rearranged. Involve only valence electrons. Associated with small energy changes.

Radioactivity
E N D
Presentation Transcript
Radioactivity First it’s here, then it’s gone!
Occur when bonds are broken and formed. Atoms remain unchanged, though they may be rearranged. Involve only valence electrons. Associated with small energy changes. Reaction rate is influenced by temperature, pressure, concentration, and catalysts. Occur when nuclei emit particles and/or rays. Atoms are often converted into atoms of another element. Involves nucleus (P/N) and electrons. Associated with large energy changes. Reaction rate is not normally affected by temperature, pressure, or catalysts. Chemical Reactions vs. Nuclear Reactions
Homework: • Read 25.1, pp 804-809 • Q pg. 809, #1, 3, 4 • Q pg. 836, #37, 38
Four forces responsible for the behavior of the particles and keep the atom together.
Strong Nuclear Force • Acts only on extremely close subatomic particles, overcomes electrostatic repulsion between positively-charged protons. • Neutrons add attractive force within nucleus. (nuclear stability is related to balance between electrostatic and strong nuclear force) • May be able to "glue" nuclei together so fusion can happen.
Electrostatic Force Combines effects of electrical charge and magnetism. Can either attract or repel particles on which it acts.
Weak Forces • Responsible for stabilizing particles through process of radioactivedecay. • Neutron changes into proton and electron.
Gravity • Force of attraction exerted between all objects in nature. • Inside tiny nucleus of atom, effect of gravity is small compared to effects of other three forces, making it weakest of all 4.
Nuclear Stability neutron-to-proton ratio • Low atomic weights (<20) • N:P ratio is 1:1 • Atoms most stable. • As atomic number increases (>20) • More N needed to produce strong nuclear force (stable nucleus) sufficient to balance electrostatic repulsion forces • N >P • N-P ratio reaches ~1.5 : 1 • No longer stable. • 20682Pb: #N / #P = 124/82 = 1.51 : 1
Stable nuclides plotted #P vs. #N, fall in band of stability. Nuclei falling outside band undergo spontaneous radioactive decay.
Types of radioactive decay On band but have > 82decay by alpha emission (too many Ns). Atoms with atomic mass above band of stability decay to increaseatomic # (too many N) Atoms with atomic mass below band decay to decrease atomic # (too many P). Depends to large degree on underlying causes for instability.
Stable nuclei tend to have even numbers of nucleons • Odd-even rule-isotopes w/even # of both N & P are far more stable than when both are odd. Of all 264 stable isotopes: • Only 5 have both odd #s of both. • 157 have even #s of both. • Rest (101) have mixed #. • Magic numbers-certain #s P or N or total nucleons, isotopes tend to be more stable (2, 8, 20, 28, 50, 82, and 126).
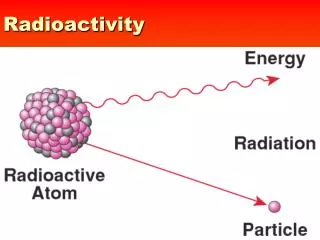
Radioactivity-particles emitted spontaneously from unstable nuclei With conflict between two strongest forces in nature, not surprising many nuclear isotopes unstable and emit some kind of radiation.
Radiation-type of penetrating rays or particles emitted by radioactive source. Radioactive decay-spontaneous process where unstable nuclei lose energy by emitting radiation. Isotopes have same atomic # but different mass #-different # neutrons. Radioisotopes-isotopes that are radioactive due to unstable nuclei.
Most common in elements w/atomic # > 82. Loses 2 N/2 P: Element changes Moves back 2 places on periodic table. Brings atom down and to left toward belt of stability nuclei. 1 relative penetrating power: Stopped by skin. Very damaging due to ionization. Alpha decay:
Nuclei w/high N : P ratio. Emission of high speed electron: N P Increases atomic # by 1. Causes shift down/to right. Releases beta particle. 100 relative penetration power: Penetrates human tissue to ~1cm. Beta emission
Most common in lighter elements w/low N : P ratio. Positron emitted (same mass as electron but opposite charge). P N Atomic # decreases by 1 but mass remains same. Causes shift up/to left. 100 relative penetrating power. Positron emission
High energy gamma ray (photon) given off when parent isotope falls into lower energy state. Neither atomic number or mass number changed, so generally not shown in nuclear equation. 10,000 relative penetrating power: Highly penetrating but not very ionizing. Gamma decay:
Common in larger elements w/low N : P ratio. Captures own inner shell electron: P N Causes shift up/to left. Reduces atomic number by 1. No release of energy or particle. Always results in gamma radiation. Electron capture
Homework: • Read 25.2, pp. 810-814 • Q pg. 814, #10-12 • Q pp. 836-837, #43, 88 a/c/f, 69-71 http://www2.wwnorton.com/college/chemistry/gilbert/tutorials/interface.swf?chapter=chapter_02&folder=nuclear_reactions http://www2.wwnorton.com/college/chemistry/gilbert/tutorials/interface.swf?chapter=chapter_02&folder=modes_of_decay
Transmutation-nucleus forms new element from disintegration of nucleus Artificial transmutation-isotope is bombarded with particle to trigger transmutation.
Radioactive decay rates • Half-life-time for ½ radioactive nuclei in any sample to decay into its products (disintegrate). • Rate of disintegration is measured in a unit called a becquerel, Bq.
2 Half-life equations: • Amount remaining = (Initial amount)(½)n • n = # half-lives that have passed • Amount remaining = (Initial amount)(½)t/T • t = elapsed time • T = duration of half-life
Volume 1 • Activity 9: What Determines and Limits an Atom’s Mass? (pp 164-185)
Radioactive dating: • Radiochemical dating-process of determining age of object by measuring amount of certain radioisotope remaining in that object. • Carbon has 7 isotopes (2 stable-others break down spontaneously): • C-10/C-11 decay by capturing electron or ejecting a positron. • C-14/-15/-16, eject electron due to large #N. • Half-life C-10 is 20 sec-because decays so fast C-10 not found in nature-observed as product of some nuclear reactions. • Half-lives of 5 unstable isotopes of C differ greatly.
C-14 produced on relatively continuous basis, and decays at steady rate with ½-life of 5730 years. Carbon-14 Dating: (not stable) N-14 + 1 n C-14 + 1 H • Over 1000s years-2 reactions reached equilibrium concentration. • Some of this C-14 reacts w/oxygen and ends up as 14CO2 -very stable. • Since ½-life of C-14 is fairly long, 14CO2 is well mixed throughout atmosphere. (As long as any organism is alive, its C atoms are being exchanged continuously with the atmosphere.) C-14 1 e- + N-14
Homework: • Read 25.3, pp. 815-820 • Q pg. 820, #21, 22, 24 • Q pp. 836-837, #51, 52, 74, 75, 77, 79, 81, 82
Nuclear Fission: • Bombard nuclei of certain isotopes w/neutrons and they undergo fission. • Splitting of nucleus into smaller fragments: • Fragments approximately equal to ½ original mass. • Sum of masses of fragments < original mass. • ~0.1% ‘missing' mass converted into energy (Einstein). • 2-3 neutrons are also emitted. • Occurs when nucleus of heavy atom captures N, or spontaneously.
Chain reaction-neutrons released in fission produce additional fission in at least one further nucleus. This nucleus, in turn, produces neutrons, and process repeats. Controlled (nuclear power). Uncontrolled (nuclear weapons). Critical mass-sample massive enough to sustain chain reaction. When present, neutrons released in one fission cause other fissions to occur. Chain Reactions
To maintain sustained controlled reaction, for every 2/3 neutrons released, only 1 must be allowed to strike another U nucleus. If <1-reaction dies out (subcritical mass): Ns escape sample before causing sufficient fissions to sustain reaction. If >1-it will grow uncontrolled (supercritical mass): chain reaction rapidly escalates, leading to violent nuclear explosion. Nuclear reactions controlled by neutron-absorbing material (graphite or cadmium): Neutron moderation-slows down Ns so they can be captured by reactor fuel to continue chain reaction. Neutron absorption-decreases # slow Ns to keep prevent chain reaction from going too quickly.
Most common uranium isotope (U-238) not suitable for nuclear weapon: • Usually neutron captured to form U-239 instead of causing fission. • U-235 has high fission probability: • Only 0.7% natural uranium is U-235. • Large amounts U needed to get necessary amount of U-235. • U-235 cannot be separated chemically from U-238, since isotopes are chemically similar. • Alternative methods developed to separate isotopes.
Release of nuclear energy by fusion of 2 light elements. H must be heated to very high Ts (100 million degrees): Needs sufficient energy to fuse (to overcome repelling forces). Then be held together long enough for fusion to occur (why called thermonuclear reactions). In hydrogen bomb, 2 isotopes of H, deuterium (1P/1N) and tritium (1P/2N), fused to form nucleus of He (2P/2N) and neutron. Releases a lot of energy. Unlike nuclear fission, there is no limit on amount of fusion that can occur. Products of fusion reaction (He and a neutron) not radioactive. Nuclear Fusion
Radiation and living things • All radiation, whether particles or waves, effects living things. • If enough energy is present, it can penetrate living cells and disrupt life processes. • Particularly dangerous if DNA/RNA molecules affected causing mutations and cancer. • When alpha or beta particles pass another atom, they pull electrons off it-atom is ionized to + ion • This slows particle down. • Why alpha particles have low penetrating power. • They ionize other atoms strongly/get slowed down. • Measurement of radiation around living things is important. • Because radioactivity affects the atoms that it passes, we can easily monitor it using variety of methods.
Henry Becquerel-1896: Uranium compounds darken (fog) photographic plates. Film badges-device containing piece of radiation-sensitive film used to monitor radiation exposure. Geiger Counter: Measures ionizing radiation. Uses ionizing effect of radioactivity. Best at detecting alpha particles that ionize strongly.
Scintillation Detectors: Phosphor-coated surface. Scintillations-bright flashes of light produced when ionizing radiation excites electrons in phosphors (NaI). Monitor nuclear power station contamination. Recognize difference between a, b and g radiation, and make different noises (such as bleeps or clicks) accordingly. Solid-State Detectors Most up-to-date instruments. Used in particle-accelerator laboratories. Show results of high-energy collisions.
Curie (Ci)-old standard unit. Equivalent to activity of 1 gram of radium. 1 curie = amount of material that will produce 3.7 x 1010 nuclear decays per second. Becquerel-more recent SI unit. 1 becquerel = amount of material which will produce 1 nuclear decay per second. 1 curie = 3.7 x 1010 becquerels. Roentgen (R) Radiation intensity of x-rays or gamma rays. Mainly used for calibration of x-ray machines. Not applicable to alpha, beta, or other particle emission. Does not accurately predict tissue effects of gamma rays of extremely high energies.
Rad Unit of absorbed radiation dose in terms of energy actually deposited in the tissue. Gray More recent SI unit. rem-roentgen-equivalent man rem = rad x Q X rays/gamma rays have Q about 1 (absorbed dose in rads is same number in rems). Neutrons have Q of about 5. Alpha particles have Q of about 20. To assess risk of radiation, absorbed dose is multiplied by relative biological effectiveness of radiation to get biological dose equivalent in rems or sieverts. sievert = 100 rems. millirem-unit of absorbed radiation dose.