Radioactivity
Radioactivity. Radioactivity. This unit explores radioactivity and how doctors can use radioactivity to diagnose and treat illnesses. Objectives. By the end of this unit you should be able to Draw diagrams of atoms Understand what an isotope is

Radioactivity
E N D
Presentation Transcript
Radioactivity The Professional Development Service for Teachers is funded by the Department of Education and Science under the National Development Plan
Radioactivity This unit explores radioactivity and how doctors can use radioactivity to diagnose and treat illnesses
Objectives By the end of this unit you should be able to • Draw diagrams of atoms • Understand what an isotope is • Understand radioactive decay and write decay equations • Understand how doctors use radioisotopes to diagnose illnesses • Understand how doctors use radioisotopes to treat cancer • Have the basic knowledge needed for Expert Group tasks
Task To help you understand this topic you will need to do some review work to find out about atoms. In your groups complete the worksheet given.
Atomic Theory Review Aim of this review: To describe the structure of the atom, state the location, relative charge, and atomic mass of the sub-atomic particles, and define atomic number and isotope. Draw the Bohr structure of some elements.
Now that you have reviewed atomic theory try using modeling clay or plasticine and pipe cleaners, make a model of (i) a lithium atom (ii) a potassium atom (iii) an aluminium atom.
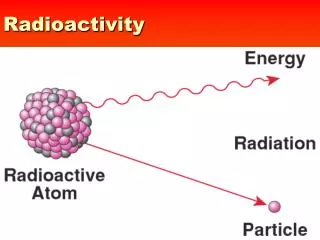
Radioactive decay Some atoms are radioactive this means their nucleus is unstable and decays to become more stable. When nuclei decay they emit or get rid of something from the nucleus
Unstable nucleus Electromagnetic wave particle Atoms which emit electromagnetic radiation or a particle by the spontaneous transformation of their nucleus are calledradioactive.
There are three types of radioactive decay Alpha decay : This is when 2 protons and 2 neutrons leave the nucleus to help it become stable Look back at the table on your review sheet what do we call something that has 2 protons and 2 neutrons in the nucleus? So an alpha particle is really ……………….nucleus If 2 protons and 2 neutrons leave the nucleus the nucleus will be transformed.
Beta Decay: This is when a neutron in the nucleus turns into a proton and electron. The electron then leaves the nucleus. Will the nucleus be transformed? Explain why
Gamma Decay: Look back at the topic on X rays .What are gamma rays? If a gamma ray leaves the nucleus will it be transformed? Explain why
- Types of radiation • Alpha Particle (α) • Beta Particle (β) • Gamma Ray (γ)
You can represent alpha and beta decay as equations :the left hand side of the arrow shows the number of protons and neutrons before the decay and the right hand side shows the number after the decay. The before and after “numbers ” must remain the same Carbon 14 written 148C is an isotope of Carbon what does this mean? It is a radioactive isotope and it decays by beta decay . The decay equation for Carbon 14 is shown below 148C→ -10e + 158N 14 particles before decay 15-1=14 particles after decay 8 protons before decay 8+0 =8 protons after decay
Problem Cobalt 60 is a radioisotope that is historically important for several reasons. It is a product in radioactive fallout from nuclear weapons. For many years, the gamma radiation from this decay was the main source for radiation therapy for cancer. Cobalt-60 decays by beta emission also. Write a decay equation to show this.
Tracers • Radioactive tracers can be used to see how well organs in your body are working or to find areas of disease. e.g. radioisotopes of iodine or technetium. • Often these are mixed with a drug that collects in a particular organ in the body. • If we then inject the drug into the body, then by detecting the radiation, we can examine that organ.
Student Activity Imagine the tubing represents the gut. There is a blockage in the gut Instead of cutting the patient to find the blockage the patient drinks a liquid containing a radioactive tracer, the dye in this model The gamma camera picks up the image of the of the liquid and the blockage is located
Gamma Camera A gamma camera detects the radiation coming from the patient and produces an image of where the radioactivity is in the body.
Obtaining the Image • The gamma camera displays the position of each gamma ray that it detects. • This is a bone scan made using technetium-99. • Can you see where the patient was injected?
Tracers and their Uses Bone :99mTc used to detect cancer, stress fractures and bone graft success. Thyroid:123 I used to access over-active and under-active thyroids, as well as thyroid tumours, cysts and goitres. Lungs:133Xe inhaled for ventilation studies (airways) and 99mTc administered for perfusion studies (blood flow). Heart:201Tl used to study muscle performance. Brain:99mTc used to assess dementia and stroke damage.Dilution analysis to access blood and water volumes (the tracer mixes with certain body fluids). Body blood: 99mTc used to assess volume. Red cells:51Cr is attached to these. Minerals:24Na and 42K used to monitor body sodium and potassium.
Radiotherapy • Rapidly dividing cells are particularly sensitive to damage by radiation. For this reason, some cancerous growths can be controlled or eliminated by irradiating the area containing the growth. External irradiation can be carried out using a gamma beam from a radioactive cobalt-60 source, • Internal radiotherapy is administered by planting a small radiation source, usually a gamma or beta emitter, in the target area. Iodine-131 is commonly used to treat thyroid cancer, probably the most successful kind of cancer treatment. It is also used to treat non-malignant thyroid disorders. Iridium-192 implants are used especially in the head and breast. They are produced in wire form and are introduced through a tube called a catheter to the target area. After administering the correct dose, the implant wire is removed to shielded storage. • Treating leukemia may involve a bone marrow transplant, in which case the defective bone marrow will first be killed off with a massive (and otherwise lethal) dose of radiation before being replaced with healthy bone marrow from a donor.
Gamma Knife This technique is used for brain surgery . Instead of opening the patient's head the gamma rays from a Cobalt 60 source are focussed on the tumour. The location of the tumour would have been accurately mapped earlier using an MRI or CT scan
The radiation must be precisely targeted. Metal windows restrict the gamma ray beam size This is the Gamma Knife in position. A polystyrene model shows the position that the patients head would occupy. A patient’s eye view
Planning the treatment A medical physicist decides how to target the gamma rays to destroy the tumour and minimise damage to other tissue